��Ŀ����
11��Ӫ��ƽ�⡢������ҩ�DZ�֤���彡����������������Ҫ;������������Ҫ��Ӫ������Ҫ�����ࡢ��֬�������ʡ����Ρ�ά���غ�ˮ�����⡢�ơ���������Ԫ���У��������������������Ԫ�ص�������
�����������У����н�����ʹ��Ч����b������ĸ����ͬ��������θ��������d��
a�������� b����˾ƥ�� c����ù�� d��������������
����ЩҩƬ���õ�����Ϊ�ϼ������۽����������������ø�Ĵ���������ˮ�⣬����ת��Ϊ�����ǣ������ʵ����ƣ���ά����C�ܷ��λ�Ѫ�������л�ԭ�ԣ���֤ά����C�л�ԭ�Կ���ʹ�õ�����Һ�͵�ˮ�����Լ����ƣ���ͬ��ɣ�
���� ��������Ҫ��Ӫ������Ҫ�����ࡢ��֬�������ʡ����Ρ�ά���غ�ˮ�����ݳ���Ԫ������Ԫ�ص������������з����жϣ�
�ڰ�˹ƥ�־��н�����ʹ��Ч�������������������ᷢ���кͷ�Ӧ��
�۵���ˮ�����������������ǣ�ά����C��ʹ��ĵ�����Һ��ɫ��ȥ��˵���ⱻ��ԭ����ά����C���л�ԭ�ԣ�
��� �⣺��������Ҫ��Ӫ������Ҫ�����ࡢ��֬�������ʡ����Ρ�ά���غ�ˮ������Ԫ�ذ�������̼���⡢�����ơ��ס��ء����ơ��ȡ�þ����Ԫ�ذ��������ܡ�ͭ��п�������̡��⡢�����⡢����������⡢�ơ���������Ԫ���У��������������������Ԫ�ص�������
�ʴ�Ϊ����֬������
�ڰ�˹ƥ�־��н�����ʹ��Ч�������������������ᷢ���кͷ�Ӧ���������θ��������������������
�ʴ�Ϊ��b��d��
�۵��۽����������������ø�Ĵ���������ˮ�⣬����ת��Ϊ������������ԣ���ĵ�����Һ��ɫ��ȥ��˵���ⱻ��ԭ����ά����C���л�ԭ�ԣ���ⷢ��������ԭ��Ӧ��ά����C��������
�ʴ�Ϊ�������ǣ���ˮ��
���� ���⿼����������Ӫ���ء�ҩ�����Ҫ�ɷֺ���Ч��֪ʶ��ע�����û�ѧ֪ʶ����������е����⣬ע�⻯ѧ����ʽ����д��Ҫ֪ʶ�㣬��Ŀ�ѶȲ���

 ȫ�ų��100��ϵ�д�
ȫ�ų��100��ϵ�д� Ӣ�ŵ��ϵ�д�
Ӣ�ŵ��ϵ�д� ������������Ծ�ϵ�д�
������������Ծ�ϵ�д�| A�� | �� | B�� | �� | C�� | �� | D�� | þ |
�ٰ��ױ�������ˮ�У���Na������CCl4�У���HF��Һ����������ƿ�У���AgNO3���屣������ɫ�Լ�ƿ�ڣ���Ũ���ᱣ������ɫ���ƿ�ڣ�
| A�� | �٢ۢ� | B�� | �ۢ� | C�� | �٢ڢۢ� | D�� | �٢ڢۢܢ� |
| A�� | Zn+CuSO4=Cu+ZnSO4 | B�� | CH4+O2=CO2+H2O | ||
| C�� | NaOH+HCl=NaCl+H2O | D�� | 2NaOH+2Al+2H2O=2NaAlO2+3H2�� |
| A�� | ��λʱ��������3n molX��ͬʱ����n mol Y | |
| B�� | X������������Z������������� | |
| C�� | X��Y��Z��Ũ�Ȳ��ٸı� | |
| D�� | X��Y��Z�ķ��Ӹ�����Ϊ3��1��2 |
| A�� | NH3+H2O?NH4++OH- | |
| B�� | HCO3-+H2O?H2CO3+OH- | |
| C�� | Cu2++2H2O?2Cu��OH��2+2H+ | |
| D�� | 2 Fe3++3H2O+3CO32-=2Fe��OH��3��+3CO2�� |
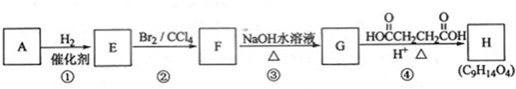
 ��
��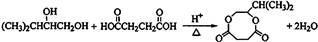 ��
��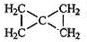 ��
��