��Ŀ����
6��ijʵ����Ҫ0.4mol•L-1NaOH��Һ480mL���ش��������⣺��1�����Ƹ���Һ��
������������ƽ����NaOH���������Ϊ8.0g��
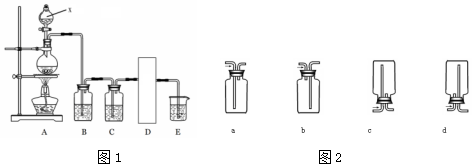
��ѡ����������ɱ�ʵ���������������������ƽ��ҩ�ס���Ͳ�⣬����Ҫ�ձ�����������500mL����ƿ����ͷ�ιܣ�
��2������ʱ����ȷ����˳����BCAFED������ţ���
A������������ˮϴ���ձ��Ͳ�����2��3�Σ�ϴ��Һ��ע������ƿ����
B���������õ�NaOH��������ձ��У��ټ�����������ˮ���ò��������裬ʹ����ȫ���ܽ�
C��������ȴ����Һ�ز�����ע����ѡ����ƿ��
D���Ǻ�ƿ�������µߵ�ҡ��
E�����ý�ͷ�ιܼ�ˮ��ʹ��Һ��Һ��ǡ����̶�������
F������������ƿ��С�ļ�ˮ��ֱ��Һ��ӽ��̶���1��2cm��
��3������A�У���ϴ��Һ��ע������ƿ����Ŀ���DZ���������ʧ��������A����������ҺŨ��ƫС���ƫ����ƫС�����䡱����
��4��ȷ��NaOH��Һ��������ݣ�ʱ�����ӹ۲�Һ��������ƿ�̶��ߣ���������ҺŨ��ƫ���ƫ����ƫС�����䡱����
��5��E�м�����ˮ���������˿̶��ߣ���������ҺŨ��ƫС���ƫ����ƫС�����䡱����Ӧ��δ����������ƣ�
���� ��1��������������Һ���ѡ���������ƿ�������m=CVM������Ҫ���ʵ�������
����������һ�����ʵ���Ũ����Һ��һ�㲽��ѡ����Ҫ��������
��2����������һ�����ʵ���Ũ����Һ��һ�㲽������
��3���������ϴ��Һ��ע������ƿ�����������ʵ���ʧ�����C=$\frac{n}{V}$��������
��4�����ӹ۲�Һ��������ƿ�̶��ߣ�������Һ���ƫС�����C=$\frac{n}{V}$��������
��5��E�м�����ˮ���������˿̶��ߣ�������Һ���ƫ���C=$\frac{n}{V}$�����������Ǵ����������ʵ��ʧ���Ҳ�����صģ������������ƣ�
��� �⣺��1������Ҫ0.4mol•L-1NaOH��Һ480mL��Ӧѡ��500mL����ƿ��ʵ������500mL��Һ����Ҫ������������m=0.4mol/L��0.5L��40g/mol=8.0g��
�ʴ�Ϊ��8.0��
���ù�������һ�����ʵ���Ũ����Һ��һ�㲽��Ϊ�����㡢�������ܽ⡢��Һ��ϴ�ӡ����ݡ�ҡ�ȵȣ��õ�����������Ϊ��������ƽ��Կ�ס��ձ�����Ͳ��������������ƿ����ͷ�ιܡ��Լ�ƿ������0.4mol•L-1NaOH��Һ480mL��Ӧѡ��500mL����ƿ�����Ի�ȱ�ٵ��������ձ�����������500mL����ƿ����ͷ�ιܣ�
�ʴ�Ϊ���ձ�����������500mL����ƿ����ͷ�ιܣ�
��2������һ�����ʵ���Ũ����Һ��һ�㲽�裺���㡢�������ܽ⡢��ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȵȣ�������ȷ�IJ���˳��Ϊ��BCAFED��
�ʴ�Ϊ��BCAFED��
��3�����ϴ��Һ��ע������ƿ�����������ʵ���ʧ������C=$\frac{n}{V}$����֪���������ҺŨ��ƫС��
�ʴ�Ϊ������������ʧ��ƫС��
��4�����ӹ۲�Һ��������ƿ�̶��ߣ�������Һ���ƫС������C=$\frac{n}{V}$��֪���������ҺŨ��ƫ��
�ʴ�Ϊ��ƫ��
��5��E�м�����ˮ���������˿̶��ߣ�������Һ���ƫ������C=$\frac{n}{V}$��֪���������ҺŨ��ƫС���ò�������ʵ��ʧ���������ȣ���Ҫ�������ƣ�
�ʴ�Ϊ��ƫС���������ƣ�
���� ���⿼����һ�����ʵ���Ũ����Һ�����ƹ����еļ���������������ڻ�������Ŀ���ѶȲ���

��.2P4+3Ba��OH��2+6H2O�T3Ba��H2PO2��2+2PH3��
��Ba��H2PO2��2+H2SO4�TBaSO4��+2H3PO2
�����ƶϲ���ȷ���ǣ�������
| A�� | ��Ӧ����������ԭ��Ӧ����Ӧ���Ƿ�������ԭ��Ӧ | |
| B�� | H3PO2���л�ԭ�ԣ��ڿ����п��ܱ����������� | |
| C�� | ��Ӧ�����������뻹ԭ��������֮��Ϊ1��1 | |
| D�� | ��Ӧ���У��ڱ�״��������2.24 L PH3ʱ��ת��0.3 mol���� |
| A�� | S��ȼ����Ϊ297KJ/mol | |
| B�� | 1mol��������ȫȼ�շų�����������297KJ | |
| C�� | 1mol��������ȫȼ�շų�������С��297KJ | |
| D�� | �γ�1mol SO2��ѧ�����ͷ����������ڶ���1molS��s����1molO2 ��g���Ļ�ѧ�������յ������� |
| ������ǽ��� | �� | �� | �� | �� | �� |
| �۵�/�� | 97.8 | 660.4 | 1535 | 1410 | 112.8 |
| �е�/�� | 883 | 2467 | 2750 | 2353 | 444.6 |
| A�� | ����� | B�� | ������ | C�� | ������ | D�� | ����� |
 MnCl2+Cl2��+2H2O��
MnCl2+Cl2��+2H2O�� ��1����ͼ��һ�ֳ����л���ı���ģ�ͣ���ģ��ͼ�ɴ���һ��D������ţ���ͬ��
��1����ͼ��һ�ֳ����л���ı���ģ�ͣ���ģ��ͼ�ɴ���һ��D������ţ���ͬ�� ��
��