��Ŀ����
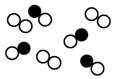
1�����������ͼ�λش��������⣺��1��ͼ��ʾ�������������ӵ�ʾ��ͼ��ͼ�С��𡱺͡��ֱ������ͬԪ�ص�ԭ�ӣ����ǵĽ����������ӣ���ͼ�пɱ�ʾ��������D���ɱ�ʾ�Ȼ��⣨HCl�����ӵ���B���ɱ�ʾһ����̼�������Ļ���������C������ţ���
 |  |  |  |
| A | B | C | D |
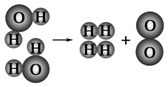
A������1mol O2��Ͽ�4mol H-O���ۼ�
B��ˮ�ֽ�����������������ķ�������Ϊ2��1
C��ˮ�ֽ�����У����ӵ������
D��ˮ�ֽ�����У�ԭ�ӵ���Ŀ����
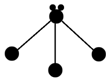
��3������ͼ�е�С�����ԭ��������1��18Ԫ�ص�ԭ��ʵ��ԭ��ʵ��ԭ�ӳ��������Ӻ�ʣ��IJ��֣���С�ڵ����δ�����γɹ��ۼ����������ӣ����ߴ������ۼ������и�ͼ��ʾ�Ľṹ�뻯ѧʽһ�����������C������ţ���
| A | B | C |
 |  | 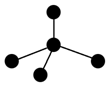 |
| NH3 | CO2 | CCl4 |
���� ��1��������ͬ��Ԫ���γɵ�˫ԭ�ӷ��ӣ�HCl�Dz�ͬԪ���γɵ�˫ԭ�ӷ��ӣ�һ����̼�Dz�ͬԪ���γɵ�˫ԭ�ӷ��ӣ�������ͬ��Ԫ���γɵ�˫ԭ�ӷ��ӣ�
��2��1molˮ�����к���2molH-O������Ӧǰ��ԭ���غ㣻
��3��A�������е�ԭ���������1�Թ¶Ե��ӣ�
B��������̼��C��O�γ�2�Թ��ۼ���
C�����Ȼ�̼��Cl����㻹��3�Թ¶Ե��ӣ�
��� �⣺��1��������ͬ��Ԫ���γɵ�˫ԭ�ӷ��ӣ�ͼ��D���ϣ�HCl�Dz�ͬԪ���γɵ�˫ԭ�ӷ��ӣ�ͼ��B���ϣ�һ����̼�Dz�ͬԪ���γɵ�˫ԭ�ӷ��ӣ�������ͬ��Ԫ���γɵ�˫ԭ�ӷ��ӣ����Ա�ʾһ����̼�������Ļ���������C��
�ʴ�Ϊ��D��B��C��
��2��A������1mol O2��2molˮ����Ͽ�4mol H-O���ۼ�����A��ȷ��
B��2molˮ���ӷֽ�����2mol������1mol��������ˮ�ֽ�����������������ķ�������Ϊ2��1����B��ȷ��
C��ˮ�ֽ�����У����ӵ�������ı䣬ԭ������䣬��C����
D���������غ��֪��ˮ�ֽ�����У�ԭ�ӵ���Ŀ���䣬��D��ȷ��
�ʴ�Ϊ��C��
��3��A��������N��H�γ����Թ��ۼ���ʣ��һ�Թ¶Ե��ӣ���A��ȷ��
B��������̼��C�ֱ�������O�γ�2�Թ��ۼ���Oʣ��2�Թ¶Ե��ӣ���B��ȷ��
C�����Ȼ�̼��Cl����������Թ¶Ե���©������C����
�ʴ�Ϊ��C��
���� ���⿼���˷��ӵ���ɡ�����Ԫ�صijɼ��ص㣬��Ŀ�ѶȲ���ע�����ԭ���γɵĹ��ۼ���Ŀ����ԭ�Ӻ���ĵ��������йأ������ڿ���ѧ���Ի���֪ʶ��Ӧ��������

| A�� | ��Ȼ�����ƾ��ֱ����ڻ�ʯ��Դ����������Դ | |
| B�� | �����ڳ�ʪ���������⣬��Ҫ�Ƿ������ⸯʴ | |
| C�� | NaClO����ǿ�����ԣ�����֯��Ư�� | |
| D�� | �ߴ���㷺Ӧ����̫���ܵ�غͰ뵼����ϵ����� |
| A�� | I2 | B�� | Si3N4 | C�� | CO2 | D�� | Si |
| A�� | ��ʽ�ζ���δ��������ϴ��ֱ��ע������� | |
| B�� | �ζ�ǰʢ��NaOH��Һ����ƿ������ˮϴ����û�и��� | |
| C�� | ��ʽ�ζ����ڵζ�ǰ�����ݣ��ζ���������ʧ | |
| D�� | ��ȡ�������ʱ����ʼ���Ӷ������ζ�����ʱ���Ӷ��� |
| A�� | 100ml 0.1mol/L����ˮ��ͨ��0.01molSO2���� | |
| B�� | 100ml 0.5mol/L��NaOH��ͨ��0.005mol���� | |
| C�� | 100ml 0.5mol/L��CuSO4��Һ�м���4g NaOH���� | |
| D�� | 100ml����ˮ�м���0.01mol�����ܽ� |
| A�� | ��ˮ������Һ̬ˮ�ͱ��У�ˮ���Ӷ��ڲ�ͣ���˶� | |
| B�� | NaCl����ˮ����Ϊ���ۼ����ƻ����Ӷ��γ���Na+��Cl- | |
| C�� | ��N2��CO2��SiO2�����У������ڹ��ۼ������Ƕ����ɷ��ӹ��� | |
| D�� | H2S�����У�����ԭ�ӵ�����㶼�ﵽ��8���ӵ��ȶ��ṹ |
 ��ͼ��ʾʵ��װ�ÿ�������ȡ��Ȳ������գ�
��ͼ��ʾʵ��װ�ÿ�������ȡ��Ȳ������գ�