��Ŀ����
A��B��C��D��E��F��G��H��I��J��Ϊ�л�������������¿�ͼ���ش���
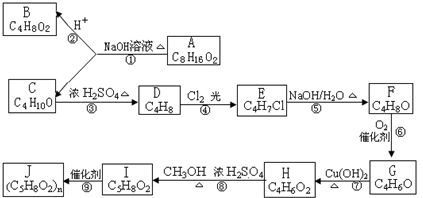
��1��B��C��Ϊ��֧�����л������B�Ľṹ��ʽΪ ��
C��Ũ���������¼��ȷ�Ӧֻ������һ��ϩ��D��D�Ľṹ��ʽΪ ��
��2��G�ܷ���������Ӧ��Ҳ��ʹ������Ȼ�̼��Һ��ɫ����G�Ľṹ��ʽΪ ��
��3��д���ݵĻ�ѧ��Ӧ����ʽ ���Ļ�ѧ��Ӧ����ʽ ����Ļ�ѧ��Ӧ����ʽ ��
��4���ٵķ�Ӧ���� ��C�й���������
��5����H������ͬ�����ŵ�H��ͬ���칹��Ľṹ��ʽΪ �� ��

��1��B��C��Ϊ��֧�����л������B�Ľṹ��ʽΪ
C��Ũ���������¼��ȷ�Ӧֻ������һ��ϩ��D��D�Ľṹ��ʽΪ
��2��G�ܷ���������Ӧ��Ҳ��ʹ������Ȼ�̼��Һ��ɫ����G�Ľṹ��ʽΪ
��3��д���ݵĻ�ѧ��Ӧ����ʽ
��4���ٵķ�Ӧ����
��5����H������ͬ�����ŵ�H��ͬ���칹��Ľṹ��ʽΪ
���㣺�л�����ƶ�
ר�⣺
����������A��B+C��ˮ�ⷴӦ�������ж�A��B��C�ֱ�����������ʹ������ɣ�1������ȷ��B��C�Ľṹ�ֱ�Ϊ��CH3��2CHCOOH�ͣ�CH3��2CHCH2OH����DΪ ��CH3��2C=CH2����D��E��ȡ����Ӧ��EΪ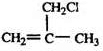 ��E����ˮ��õ���FΪCH2=C��CH3��CH2OH��F�����õ�GΪCH2=C��CH3��-CHO��������Ӧ�ߵõ�����HΪCH2=C��CH3��-COOH��H��CH3OH�õ���IΪCH2=C��CH3��-COOCH3����JΪ�Ӿ۷�Ӧ�IJ��Ϊ
��E����ˮ��õ���FΪCH2=C��CH3��CH2OH��F�����õ�GΪCH2=C��CH3��-CHO��������Ӧ�ߵõ�����HΪCH2=C��CH3��-COOH��H��CH3OH�õ���IΪCH2=C��CH3��-COOCH3����JΪ�Ӿ۷�Ӧ�IJ��Ϊ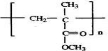 ���ݴ˴��⣻
���ݴ˴��⣻
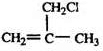 ��E����ˮ��õ���FΪCH2=C��CH3��CH2OH��F�����õ�GΪCH2=C��CH3��-CHO��������Ӧ�ߵõ�����HΪCH2=C��CH3��-COOH��H��CH3OH�õ���IΪCH2=C��CH3��-COOCH3����JΪ�Ӿ۷�Ӧ�IJ��Ϊ
��E����ˮ��õ���FΪCH2=C��CH3��CH2OH��F�����õ�GΪCH2=C��CH3��-CHO��������Ӧ�ߵõ�����HΪCH2=C��CH3��-COOH��H��CH3OH�õ���IΪCH2=C��CH3��-COOCH3����JΪ�Ӿ۷�Ӧ�IJ��Ϊ ���ݴ˴��⣻
���ݴ˴��⣻���
�⣺����A��B+C��ˮ�ⷴӦ�������ж�A��B��C�ֱ�����������ʹ������ɣ�1������ȷ��B��C�Ľṹ�ֱ�Ϊ��CH3��2CHCOOH�ͣ�CH3��2CHCH2OH����DΪ ��CH3��2C=CH2����D��E��ȡ����Ӧ��EΪ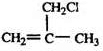 ��E����ˮ��õ���FΪCH2=C��CH3��CH2OH��F�����õ�GΪCH2=C��CH3��-CHO��������Ӧ�ߵõ�����HΪCH2=C��CH3��-COOH��H��CH3OH�õ���IΪCH2=C��CH3��-COOCH3����JΪ�Ӿ۷�Ӧ�IJ��Ϊ
��E����ˮ��õ���FΪCH2=C��CH3��CH2OH��F�����õ�GΪCH2=C��CH3��-CHO��������Ӧ�ߵõ�����HΪCH2=C��CH3��-COOH��H��CH3OH�õ���IΪCH2=C��CH3��-COOCH3����JΪ�Ӿ۷�Ӧ�IJ��Ϊ ��
��
��1�������Ϸ�����֪BΪ��CH3��2CHCOOH��DΪ��CH3��2C�TCH2��
�ʴ�Ϊ����CH3��2CHCOOH����CH3��2C�TCH2��
��2�������Ϸ�����֪GΪCH2=C��CH3��-CHO��
�ʴ�Ϊ��CH2=C��CH3��-CHO��
��3����Ӧ��Ϊ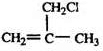 ��ˮ�ⷴӦ����Ӧ�ķ���ʽΪ
��ˮ�ⷴӦ����Ӧ�ķ���ʽΪ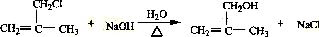 ����Ӧ��ΪCH2=C��CH3��CH2OH�����õ�CH2=C��CH3��-CHO����Ӧ�Ļ�ѧ��Ӧ����ʽΪ2CH2=C��CH3��CH2OH+O2
����Ӧ��ΪCH2=C��CH3��CH2OH�����õ�CH2=C��CH3��-CHO����Ӧ�Ļ�ѧ��Ӧ����ʽΪ2CH2=C��CH3��CH2OH+O2
2CH2=C��CH3��-CHO+2H2O����Ӧ��ΪCH2=C��CH3��-COOCH3�ļӾ۷�Ӧ����Ӧ�ķ���ʽΪ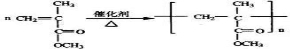 ��
��
�ʴ�Ϊ��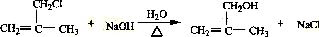 ��2CH2=C��CH3��CH2OH+O2
��2CH2=C��CH3��CH2OH+O2
2CH2=C��CH3��-CHO+2H2O�� ��
��
��4���ɷ�Ӧ���������ŵı仯��֪��Ӧ��Ϊˮ�ⷴӦ��CΪ��CH3��2CHCH2OH�����еĹ�����Ϊ�ǻ���
�ʴ�Ϊ��ˮ�ⷴӦ���ǻ���
��5��HΪCH2=C��CH3��-COOH����H������ͬ�����ŵ�H��ͬ���칹����CH2=CHCH2COOH ��CH3CH=CHCOOH��
�ʴ�Ϊ��CH2=CHCH2COOH��CH3CH=CHCOOH��
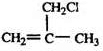 ��E����ˮ��õ���FΪCH2=C��CH3��CH2OH��F�����õ�GΪCH2=C��CH3��-CHO��������Ӧ�ߵõ�����HΪCH2=C��CH3��-COOH��H��CH3OH�õ���IΪCH2=C��CH3��-COOCH3����JΪ�Ӿ۷�Ӧ�IJ��Ϊ
��E����ˮ��õ���FΪCH2=C��CH3��CH2OH��F�����õ�GΪCH2=C��CH3��-CHO��������Ӧ�ߵõ�����HΪCH2=C��CH3��-COOH��H��CH3OH�õ���IΪCH2=C��CH3��-COOCH3����JΪ�Ӿ۷�Ӧ�IJ��Ϊ ��
����1�������Ϸ�����֪BΪ��CH3��2CHCOOH��DΪ��CH3��2C�TCH2��
�ʴ�Ϊ����CH3��2CHCOOH����CH3��2C�TCH2��
��2�������Ϸ�����֪GΪCH2=C��CH3��-CHO��
�ʴ�Ϊ��CH2=C��CH3��-CHO��
��3����Ӧ��Ϊ
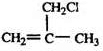 ��ˮ�ⷴӦ����Ӧ�ķ���ʽΪ
��ˮ�ⷴӦ����Ӧ�ķ���ʽΪ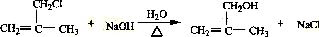 ����Ӧ��ΪCH2=C��CH3��CH2OH�����õ�CH2=C��CH3��-CHO����Ӧ�Ļ�ѧ��Ӧ����ʽΪ2CH2=C��CH3��CH2OH+O2
����Ӧ��ΪCH2=C��CH3��CH2OH�����õ�CH2=C��CH3��-CHO����Ӧ�Ļ�ѧ��Ӧ����ʽΪ2CH2=C��CH3��CH2OH+O2| ���� |
 ��
���ʴ�Ϊ��
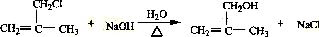 ��2CH2=C��CH3��CH2OH+O2
��2CH2=C��CH3��CH2OH+O2| ���� |
 ��
����4���ɷ�Ӧ���������ŵı仯��֪��Ӧ��Ϊˮ�ⷴӦ��CΪ��CH3��2CHCH2OH�����еĹ�����Ϊ�ǻ���
�ʴ�Ϊ��ˮ�ⷴӦ���ǻ���
��5��HΪCH2=C��CH3��-COOH����H������ͬ�����ŵ�H��ͬ���칹����CH2=CHCH2COOH ��CH3CH=CHCOOH��
�ʴ�Ϊ��CH2=CHCH2COOH��CH3CH=CHCOOH��
���������⿼���л�����ƶϣ����ؼ����ҽ����ͻ�ƿڣ������ۣ�������A��B+C��ˮ�ⷴӦ�������ж�A��B��C�ֱ�����������ʹ������ɣ�1������ȷ��B��C�Ľṹ���Դ˿��ƶ��������ʣ�ע���л�������ŵĽṹ�����ʣ�Ϊ��ȷ��������Ŀ�Ĺؼ�����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
���¹���ʵ�������˵����ȷ���ǣ�������
| A������ʱΪ�ӿ��ٶȿ����ò����������� |
| B������ʱ���������ʯ������ |
| C�����þƾ���ȡ��ˮ�еĵ� |
| D������ʱ�¶ȼ�Һ��Ӧ��������ƿ֧�ܿ�ƽ�� |
ѡ��������Ũ����ķ�Ӧ������ߵ�ϩ����������
A�� |
B��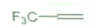 |
| C���T |
���д��á��������������������ȷ���ǣ�������
��Ư��¶���ڿ����б���
�ڽ���þ�Ż�ʱ������CO2���
����һ����ͨ�����壬�����һ�������ġ�ͨ·��
���д���������й©ʱ��Ӧ�ý�����������Һ��ë����ס�ڱ���ߴ��ܣ�
��Ư��¶���ڿ����б���
�ڽ���þ�Ż�ʱ������CO2���
����һ����ͨ�����壬�����һ�������ġ�ͨ·��
���д���������й©ʱ��Ӧ�ý�����������Һ��ë����ס�ڱ���ߴ��ܣ�
| A���٢� | B���٢� | C���ڢ� | D���ۢ� |
ʳ�Ρ����ʳ��Ϊ�����г��õ����ʣ�������Щ���ʲ���ʵ�ֵ�ʵ���ǣ�������
�ٳ�ȥ��ˮƿ�ڱڵ�ˮ��
�ڼ�������ˮ���Ƿ���������
�ۼ���þ�����Ƿ������ۡ���
�ܼ���ʳ�κʹ��
�ٳ�ȥ��ˮƿ�ڱڵ�ˮ��
�ڼ�������ˮ���Ƿ���������
�ۼ���þ�����Ƿ������ۡ���
�ܼ���ʳ�κʹ��
| A���٢� | B���ڢ� | C���ڢ� | D���٢� |
�����£�������Һ�е���Ũ�ȹ�ϵ��ȷ���ǣ�������
| A������ͨ������������Һ�У�[Na+]=[Cl-]+[ClO-]+[OH-] |
| B��pH=8.3��NaHCO3��Һ��[Na+]��[HCO3-]��[CO32-]��[H2CO3] |
| C��pH=11�İ�ˮ��pH=3������������ϣ�[Cl-]=[NH4+]��[OH-]=[H+] |
| D��ij��Һ��ֻ����SO42-��OH-��NH4+��H+�������ӣ�����ܴ��ڣ�[NH4+]��[SO42-]��[H+]��[OH-] |
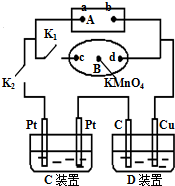 ��1����ͼ��ʾ��AΪ��Դ��BΪ������ʳ��ˮ�ͷ�̪��Һ����ֽ����ֽ�������һ��KMnO4��Һ��C��DΪ���ۣ���缫���ϼ�ͼ���������Һ����֪��
��1����ͼ��ʾ��AΪ��Դ��BΪ������ʳ��ˮ�ͷ�̪��Һ����ֽ����ֽ�������һ��KMnO4��Һ��C��DΪ���ۣ���缫���ϼ�ͼ���������Һ����֪��