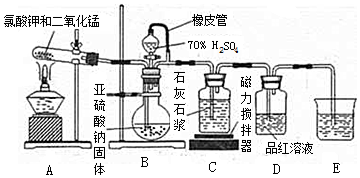
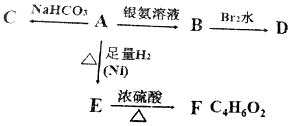
��Ŀ����
12�������г�����Ca2+��Mg2+��SO42-�ȿ����������⣬��������ɰ�Ȳ��������ʣ���1��������Һ�к���SO42-�ķ�����ȡ������Һ���Թ��У��μ��������ᣬ�ٵμӼ���BaCl2��Һ���а�ɫ�������ɣ�˵����Һ�к���SO42-��
��2���������ܽ���˳�ȥ���������ʺ�����Һ�����εμӹ���NaOH��Һ��BaCl2��Һ��Na2CO3��Һ��ֱ�����ٲ�����������Щ������Ŀ�������γ�ȥMg2+��SO42-��Ca2+������Ba2+���μ�Na2CO3��Һʱ������Ӧ�Ļ�ѧ����ʽΪBaCl2+Na2CO3�TBaCl2��+2NaCl��CaCl2+Na2CO3�TCaCO3��+2NaCl��
���� ��1��ѡ���ᡢ�Ȼ���������������ӣ��ȼ������ų��������ӵĸ��ţ�
��2��ֱ�����ٲ����������ɽ�������ȫת��Ϊ��������̼���ƿɳ�ȥ�����ı����ӣ��Դ������
��� �⣺��1��������Һ�к���SO42-�ķ�����ȡ������Һ���Թ��У��μ��������ᣬ�ٵμӼ���BaCl2��Һ���а�ɫ�������ɣ�˵����Һ�к���SO42-��
�ʴ�Ϊ��ȡ������Һ���Թ��У��μ��������ᣬ�ٵμӼ���BaCl2��Һ���а�ɫ�������ɣ�˵����Һ�к���SO42-��
��2���������ܽ���˳�ȥ���������ʺ�����Һ�����εμӹ���NaOH��Һ��BaCl2��Һ��Na2CO3��Һ��ֱ�����ٲ�����������Щ������Ŀ�������γ�ȥMg2+��SO42-��Ca2+������Ba2+���μ�Na2CO3��Һʱ������Ӧ�Ļ�ѧ����ʽΪBaCl2+Na2CO3�TBaCl2��+2NaCl��CaCl2+Na2CO3�TCaCO3��+2NaCl��
�ʴ�Ϊ�����γ�ȥMg2+��SO42-��Ca2+������Ba2+��BaCl2+Na2CO3�TBaCl2��+2NaCl��CaCl2+Na2CO3�TCaCO3��+2NaCl��
���� ���⿼������ᴿ��Ϊ��Ƶ���㣬���ճ����Լ���ѡ���Ⱥ�˳����ԭ��Ϊ���Ĺؼ������ط�����ʵ�������Ŀ��飬ע��̼�������Ȼ���֮����Ŀ�ѶȲ���

| A�� | Na+��I-��K+��Br2 | B�� | Mg2+��Cu2+��SO42-��NO3- | ||
| C�� | Fe2+��Cl2��K+��SO42- | D�� | ClO-��Cl-��CO2��Na+ |
| A�� | dacb | B�� | bcad | C�� | acbd | D�� | dcba |
| A�� | ���ȵ��ռ���Һ��ȥ���������۹� | |
| B�� | ���Ʋ;߲��˳�ʱ�������ԡ����Ի��̵����� | |
| C�� | Ư�۳��ڷ��ÿ����лᱻ�����е��������������� | |
| D�� | ����β���к��еĵ�����������Ͳ���ȫȼ����ɵ� |
| A�� | AgNO3 | B�� | NaOH | C�� | Na2CO3 | D�� | AlCl3 |