��Ŀ����
16���̷����壨FeSO4•7H2O��M=278g/mol��������ȱ����ƶѪҩƷ����Ҫ�ɷ֣�ʵ�����������᳧����������Ҫ�ɷ�ΪFe2O3������FeS��SiO2�����Ʊ��̷��Ĺ������£�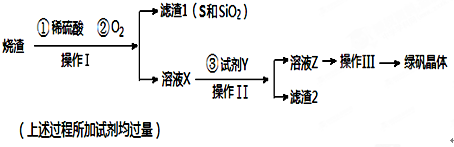
�Իش�
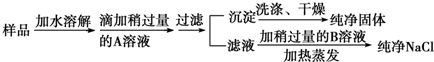
��1������ IΪ���ˣ���д�������ƣ���
��2���Լ�Y����ҺX��Ӧ�����ӷ���ʽΪFe+2Fe3+�T3Fe2+��Fe+2H+�TFe2++H2��
��3������ III��˳������Ϊ������Ũ������ȴ�ᾧ�����ˡ�ϴ�ӡ����
��4��ijͬѧ������KMnO4��Һ�ⶨ�̷���Ʒ��Fe2+������
a����ȡ11.5g�̷���Ʒ���ܽ⣬���Ƴ�1000mL��Һ��
b����ȡ25.00mL������Һ����ƿ�У�
c���������ữ��0.01000mol/L KMnO4��Һ�ζ����յ㣬����KMnO4��Һ�������ƽ��ֵΪ20.00mL��
�ٲ���a������Һʱ��Ҫ�IJ�������������������Ͳ���ձ�����ͷ�ι��⣬����1000mL����ƿ
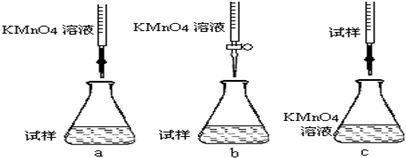
�ڸ�ͬѧ��Ƶ����еζ���ʽ�����������b���гֲ�����ȥ��������ĸ��ţ�
���жϴ˵ζ�ʵ��ﵽ�յ�ķ����ǵμ����һ��KMnO4��Һʱ����Һ�����ɫ�Ұ���Ӳ�����ɫ�����ڵζ��յ��ȡ�ζ��̶ܿ�ʱ������KMnO4��ҺҺ�棬������������ȷ����ʹ�ⶨ���ƫ�ͣ��ƫ�ߡ���ƫ�͡�����Ӱ�족����
�ܼ���������Ʒ��FeSO4•7H2O����������Ϊ96.7%��
���� ���᳧���������ᡢ������Ӧ�����˷���õ���ҺX�Ͳ�����I��S����������ȣ�����ҺX��ҪΪFe2��SO4��3��H2SO4�ȣ�����������������������Һ�����ɳ�ȥδ��Ӧ�����ᣬ���˳�ȥδ��Ӧ��Fe����ҺΪ����������Һ����ͨ��Ũ����������ȴ�ᾧ�õ��̷���
��1�����ղ�����ǰ������ʱ仯����֪�������ǹ��ˣ�
��2������Fe��Fe3+��ԭΪFe2+�����ɳ�ȥδ��Ӧ�����
��3���������Ǵ���Һ�л�þ��壻
��4����������Һ����ȻҪ�õ�����ƿ��Ӧ��ע������ƿ�Ĺ��
�ڸ��������Һ����ǿ�����ԣ��������ܣ�Ӧ������ʽ�ζ��ܣ�ע�����������������
�۸��������Һ�������Ϻ�ɫ����������ָʾ����������Һ��ɫ�ж��յ㣻�ζ��յ��ȡ�ζ��̶ܿ�ʱ������KMnO4��ҺҺ�棬Һ���ڿ̶����·����������������Һ�����ƫС��
�ܸ������ĸ�����ؼ���n��Fe2+������n��FeSO4•7H2O��=n��Fe2+��������������Ʒ��FeSO4•7H2O������������
��� �⣺���᳧���������ᡢ������Ӧ�����˷���õ���ҺX�Ͳ�����I��S����������ȣ�����ҺX��ҪΪFe2��SO4��3��H2SO4�ȣ�����������������������Һ�����ɳ�ȥδ��Ӧ�����ᣬ���˳�ȥδ��Ӧ��Fe����ҺΪ����������Һ����ͨ��Ũ����������ȴ�ᾧ�õ��̷���
��1���������ǽ��������ܵĹ�����Һ����룬��֪�������ǹ��ˣ�
�ʴ�Ϊ�����ˣ�
��2������Fe��Fe3+��ԭΪFe2+�����ɳ�ȥδ��Ӧ�����ᣬ��Ӧ���ӷ���ʽΪ��Fe+2Fe3+�T3Fe2+��Fe+2H+�TFe2++H2����
�ʴ�Ϊ��Fe+2Fe3+�T3Fe2+��Fe+2H+�TFe2++H2����
��3���������Ǵ���Һ�л�þ��壬��������Ũ������ȴ�ᾧ�����ˡ�ϴ�ӡ�����õ����壬
�ʴ�Ϊ������Ũ����
��4����������Һ����ȻҪ�õ�����ƿ����Ҫ1000mL����ƿ��
�ʴ�Ϊ��1000mL����ƿ��
�ڸ��������Һ����ǿ�����ԣ��������ܣ�Ӧ������ʽ�ζ��ܣ�cװ��������Ӵ��Ƚϴ��ױ�������������ѡ��b��
�۸��������Һ�������Ϻ�ɫ����������ָʾ�����μ����һ��KMnO4��Һʱ����Һ�����ɫ�Ұ���Ӳ�����ɫ��˵����Ӧ�����յ㣬
�ζ��յ��ȡ�ζ��̶ܿ�ʱ������KMnO4��ҺҺ�棬Һ���ڿ̶����·����������������Һ�����ƫС���ⶨ���ƫ�ͣ�
�ʴ�Ϊ���μ����һ��KMnO4��Һʱ����Һ�����ɫ�Ұ���Ӳ�����ɫ��ƫ�ͣ�
��25mL��Һ���ĸ������Ϊ0.02L��0.01mol/L=0.0002mol����5Fe2++MnO4-+8H+�T5Fe3++Mn2++4H2O����֪11.5g�̷���Ʒ��n��Fe2+��=5��0.0002mol��$\frac{1000mL}{25mL}$=0.04mol����n��FeSO4•7H2O��=n��Fe2+��=0.004mol����Ʒ��FeSO4•7H2O����������Ϊ$\frac{0.04mol��278g/mol}{11.5g}$��100%=96.7%��
�ʴ�Ϊ��96.7%��
���� ���⿼�����ʵ��Ʊ���Ϊ�߿��������ͣ��漰���ʵķ����ᴿ����Һ���ơ�������ԭ��Ӧ�ζ������ʺ����ⶨ�ȣ���Ŀ�Ѷ��еȣ��ؼ�����ȷ�Ʊ�ԭ������ѧʵ�����������

��1������Ҫ�ƾ��ơ�������������ǯ��ҩ�ס����żܵ������⣬������������ѡ�������������ñ����ĸ��д��ABDF��
A��������ƽ B���в� C���Թܼ� D������ E�������� F�������� G��ʯ����
����Ҫ�������������ǣ�
��2��ijѧ��ʵ���õ��������ݣ�
| ����ǰ������ | ���Ⱥ������ | ||
| W1�������� | W2������+���壩 | W3������+��ˮ����ͭ�� | |
| ��һ�� | 15.688g | 17.668g | 16.962g |
| �ڶ��� | 15.688g | 17.744g | 17.033g |
��3����ѧ���ⶨ�����ƫ����ƫ�ͣ�ƫ�ͣ�������ѡ����ѡ����ѧ����������ԭ������ǣ���д��ĸ��BC��
A������ǰ����ʱ����δ��ȫ�����B��������μ��Ⱥ���������ϴ���0.001g��
C�����Ⱥ�����δ�������������ȴ��D�����ȹ����о�����������ʧ��
��4���ⶨ����ͭ�����нᾧˮ�ĺ�������������ƽ�Ͻ����Ĵγ������������Ĵγ�����Ŀ���ǽ��к��ز������жϴﵽ���ص�������ֱ���������γ������ó���0.001gΪֹ��
| A�� | �ݢܢ٢ۢ� | B�� | �ܢ٢ڢݢ� | C�� | �ڢݢܢ٢� | D�� | �٢ܢڢݢ� |
| A�� | Cl | B�� | O | C�� | KMnO4 | D�� | HCl |