��Ŀ����
9��ijҺ������ֻ������K+��NH4+��NO3-��SO42-��Cl-��Cu2+�е�һ�ֻ���������ɣ����ν�������ʵ�飬�۲쵽��ʵ�������¼���£��ٸû��Һ��Ϊ��ɫ��Һ��ȡ�������Թ��У�����NaOH��Һ�����ȣ���ʪ��ĺ�ɫʯ����ֽ�����Թܿڣ�δ�۲쵽��ֽ������
���øɾ��IJ�˿պȡ������ڻ��������գ�����ɫ���ܲ��������Թ۲쵽�������ɫ��
����������Һ�еμӹ���BaCl2��Һ���а�ɫ�������ɣ�
����۵���Һ�м���AgNO3��Һ���а�ɫ�������ɣ��ó���������ϡ���ᣮ
�ݴˣ����жϻ�����п϶�����K+��SO42-���϶�������Cu2+��NH4+�����ܿ϶��Ƿ���Cl-��NO3-��
���� �ٸû��Һ��Ϊ��ɫ��Һ��˵��û��Cu2+��ȡ�������Թ��У�����NaOH��Һ�����ȣ���ʪ��ĺ�ɫʯ����ֽ�����Թܿڣ�δ�۲쵽��ֽ������˵����Һ����NH4+��
���øɾ��IJ�˿պȡ������ڻ��������գ�����ɫ���ܲ��������Թ۲쵽�������ɫ����֪��Һ����K+��
����������Һ�еμӹ���BaCl2��Һ���а�ɫ�������ɣ��˳���ΪBaSO4����Һ����SO42-��
����۵���Һ�м���AgNO3��Һ���а�ɫ�������ɣ��ó���������ϡ���ᣬ�˳���ΪAgCl����������������Ȼ�����Һ������Cl-������ȷ����ԭ��Һ���Cl-��
��� �⣺�ٸû��Һ��Ϊ��ɫ��Һ��˵��û��Cu2+��ȡ�������Թ��У�����NaOH��Һ�����ȣ���ʪ��ĺ�ɫʯ����ֽ�����Թܿڣ�δ�۲쵽��ֽ������˵����Һ����NH4+��
���øɾ��IJ�˿պȡ������ڻ��������գ�����ɫ���ܲ��������Թ۲쵽�������ɫ����֪��Һ����K+��
����������Һ�еμӹ���BaCl2��Һ���а�ɫ�������ɣ��˳���ΪBaSO4����Һ����SO42-��
����۵���Һ�м���AgNO3��Һ���а�ɫ�������ɣ��ó���������ϡ���ᣬ�˳���ΪAgCl����������������Ȼ�����Һ������Cl-������ȷ����ԭ��Һ���Cl-��
�������Ϸ�����ԭ��Һ��һ����K+��SO42-��һ��û��Cu2+��NH4+��������Cl-��NO3-���ʴ�Ϊ��K+��SO42-��Cu2+��NH4+��Cl-��NO3-��
���� ���⿼�����ӵļ��鼰���ӹ��棬���ճ������ӵļ��鼰�����Ļ�ѧ��ӦΪ���Ĺؼ�������ѧ������������˼ά�����ԵĿ��飬��Ŀ�Ѷ��еȣ�

| A�� | 7.8g Na2O2�к��е���������ĿΪ0.2 NA | |
| B�� | 0.1mol Fe������ϡHNO3��Ӧ��ת�Ƶ�����Ϊ0.3NA | |
| C�� | 1L 0.1 mol/L Al2��SO4��3��Һ�У�Al3+����ĿΪ0.2 NA | |
| D�� | ��״���£�2.24L CHCl3�ķ�����Ϊ0.1 NA |
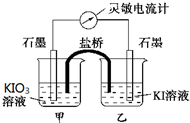 ����ӦIO3-+5I-+6H+?3I2+3H2O��Ƴ���ͼ��ʾ��ԭ��أ��ס����ձ��ж����������Һ����ʼʱ����ձ��м�������Ũ���ᣬ������ָ�뷢��ƫת��һ��ʱ�������ָ��ص��㣬������ձ��е��뼸��ŨNaOH��Һ��������ָ���ٴη���ƫת�������жϲ���ȷ���ǣ�������
����ӦIO3-+5I-+6H+?3I2+3H2O��Ƴ���ͼ��ʾ��ԭ��أ��ס����ձ��ж����������Һ����ʼʱ����ձ��м�������Ũ���ᣬ������ָ�뷢��ƫת��һ��ʱ�������ָ��ص��㣬������ձ��е��뼸��ŨNaOH��Һ��������ָ���ٴη���ƫת�������жϲ���ȷ���ǣ�������| A�� | ���ε�����ָ��ƫת�����෴�������ƶ���Ϊ��ʱ����Ӧ�ﵽ��ѧƽ��״̬ | |
| B�� | ����ʵ���У������е��������ƶ������෴ | |
| C�� | ��ʼ��������Ũ����ʱ��ֻ�����ձ�����Һ���� | |
| D�� | ����ձ��е��뼸��ŨNaOH��Һ������ʯī�缫�Ϸ�����ԭ��Ӧ |
| ������ | K+ NH4+ Fe2+ Mg2+ Al3+ Cu2+ |
| ������ | OH- Cl- Al02- CO32- SiO32-SO42-�� |
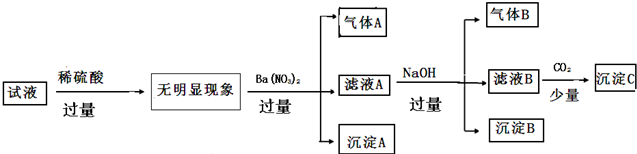
����˵����ȷ���ǣ�������
| A�� | ԭ��Һ��ֻ����NH4+ Fe2+Cl- SO42- | |
| B�� | �ɳ���A�ƶ�ԭ��Һ��һ������SO42- | |
| C�� | ��ҺA�п��ܺ���K+ Al3+ Cl- | |
| D�� | ����B��һ������Mg��OH��2 |
| A�� | ������Ҷ��� | B�� | ���Ѻ��Ҵ� | C�� | ��ȩ������ | D�� | ��ȩ�ͱ�ͪ |
| A�� | HCl | B�� | CH3COO- | C�� | SO42- | D�� | Fe3+ |