��Ŀ����
9������������ȷ���ǣ���������1��c��H+���Tc��OH-��=10-6mol/L����Һһ��������
��2����֪NaOH��aq��+HCl��aq���TNaCl��aq��+H2O��l����H=-57.3kJ•mol-1����40.0g NaOH��ϡ��Һ��ϡ������ȫ�кͣ��ų�57.3kJ������
��3���������Һ�м���ϡH2SO4�����ȼ����ӣ���ȴ���ټ�������Cu��OH��2����Һ�����ȣ���ש��ɫ�������ɣ�˵������û��ˮ������������
��4��ͬһ�����Ԫ�أ�ԭ�Ӱ뾶Խ���䵥�ʵ��۵㲻һ��Խ��
��5������������NaOH��Һ��H218O����ˮ�����ΪCH3CO18ONa��CH3CH2OH
��6��ˮ�ĵ���ʽ H+[��O��]2-H+��
| A�� | ��1����2����5����6�� | B�� | ��1����4����5�� | C�� | ��2����4����6�� | D�� | ȫ�� |
���� ��1����Һ�Ƿ������ȡ����c��H+����c��OH-���Ĵ�С��
��2������Ϊ���ᣬ�������ȣ�
��3����������������ͭ��Һ�ķ�Ӧ�ڼ��������½��У�
��4��ͬһ�����Ԫ�أ���Ϊ�������ʣ�ԭ�Ӱ뾶Խ���ʵ��۵�Խ�ͣ�
��5��������Ҵ�����������Ӧʱ�������ǻ������⡱������������ˮ��Ϊ������Ӧ���淴Ӧ��
��6��ˮΪ���ۻ����
��� �⣺��1����Һ�Ƿ������ȡ����c��H+����c��OH-���Ĵ�С��ֻҪ����c��H+���Tc��OH-��������Һ�����ԣ�����ȷ��
��2������Ϊ���ᣬ�������ȣ���40.0g NaOH��ϡ��Һ��ϡ������ȫ�кͣ��ų�����С��57.3kJ���ʴ���
��3���������ذ���������ˮ�⣬����������������ͭ��Һ�ķ�Ӧ�ڼ��������½��У�ˮ���Ӧ�ȼ�����������ʱ��Һ��Ϊ���ԣ��ʴ���
��4��ͬһ�����Ԫ�أ���Ϊ�������ʣ�ԭ�Ӱ뾶Խ���ʵ��۵�Խ�ͣ�����ȷ��
��5�������������������Ƶ�H218O��ˮ������CH3CO18ONa��C2H5OH������ȷ��
��6��ˮΪ���ۻ��������ʽΪ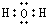 ���ʴ���
���ʴ���
��ѡB��
���� ���⿼���Ϊ�ۺϣ��漰������ʵĵ��롢���۵�ˮ�⡢Ԫ�������ʡ����������������Լ�����ʽ����д��֪ʶ��Ϊ��Ƶ���㣬������˫���Ŀ��飬ע����ػ���֪ʶ��ѧϰ���ѶȲ���

 �ִʾ��ƪϵ�д�
�ִʾ��ƪϵ�д�| A�� | �÷�Ӧ��Al�ǻ�ԭ����ϡ������������ | |
| B�� | �÷�Ӧ������Al������Ϊ1.8g | |
| C�� | ����2.24L H2 | |
| D�� | �÷�Ӧ����0.3NA�����ӷ���ת�� |
| A�� | HCl | B�� | NO | C�� | NH3 | D�� | Cl2 |
| A�� | ����CO2ͨ��NaOH��Һ�У�CO2+2OH-�TCO32-+H2O | |
| B�� | ͭ������Ũ���ᷴӦ��Cu+4H++2NO3-�TCu2++2NO��+2H2O | |
| C�� | ̼���ƹ�������ˮ��CO32-+2H2O?H2CO3+2OH- | |
| D�� | ����ˮ��Ӧ��2Na+2H2O�T2Na++2OH-+H2�� |
| A�� | Ħ���ǹ��ʵ�λ�Ƶ��߸�������֮һ | |
| B�� | Ħ���DZ�ʾ���������ĵ�λ | |
| C�� | ��ѧ�Ϲ涨���а����ӵ����������ӵ��κ����Ӽ�����Ϊ1mol | |
| D�� | 1mol����6.02��1023��O2 |
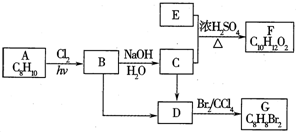 A��G�����л���������ǵ�ת����ϵ���£�
A��G�����л���������ǵ�ת����ϵ���£�
 ��
�� ���ṹ��ʽ��
���ṹ��ʽ��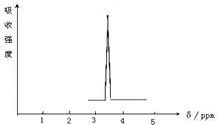 ��Ҫ������������⣺
��Ҫ������������⣺