��Ŀ����
С��ͬѧ����CuSO4?5H2O����480mL 1mol/L����Һ��
��1��С��ͬѧѡ�õ�����ƿ�ݻ��Ĺ���� mL��
��2���������ڸ�ʵ��������Ҫ����;���ֱ��� �� ��
��3��С��ͬѧͨ�����㣬������ȡ g CuSO4?5H2O��
��4�����ƺõ���Һ�� ɫ��С��ͬѧ����ͨ��ʵ��ֱ�֤ʵ�����к���Cu2+��SO42-�������������취��ѡ���ʵ����Լ���д��ѧʽ������д������Ӧ�����ӷ���ʽ��
��5�����ʵ���Ũ��������������ƫ�ߡ�ƫ�͡���Ӱ�죩
��������ƿ��ϴ����δ�����������ˮ���������Ƶ���ҺŨ�Ƚ� ��
�ڶ���ʱ�����۾����ӣ��������Ƶ���ҺŨ�Ƚ� ��
�۶���ʱ��Һ�泬���̶��ߣ���������ҺŨ�Ƚ� ��
��1��С��ͬѧѡ�õ�����ƿ�ݻ��Ĺ����
��2���������ڸ�ʵ��������Ҫ����;���ֱ���
��3��С��ͬѧͨ�����㣬������ȡ
��4�����ƺõ���Һ��
| ���� | �����Լ� | ���ӷ���ʽ |
| Cu2+ | ||
| SO42- |
��������ƿ��ϴ����δ�����������ˮ���������Ƶ���ҺŨ�Ƚ�
�ڶ���ʱ�����۾����ӣ��������Ƶ���ҺŨ�Ƚ�
�۶���ʱ��Һ�泬���̶��ߣ���������ҺŨ�Ƚ�
���㣺����һ�����ʵ���Ũ�ȵ���Һ
ר�⣺
��������1������ʵ���ҳ�������ƿ�Ĺ��ѡ��
��2���ܽ��ת��ʹ�ò�������
��3������n=c��V��m=n��M��������Ҫ��CuSO4?5H2O��������
��4��ͭ���ӳ���ɫ��ͭ���Ӽ��飺����Һ�м����������ƣ�����ɫ�������ɣ���������ӵļ��飺����Һ�м�������ʹ֮�ữ���ټ���BaCl2��Һ���а�ɫ����������
��5�����ʵ���Ũ����������������Ũ�ȹ�ʽC=
������ʹ��������ʵ����ʵ���ƫ�����ʹ��Һ�����ƫС�IJ���������ʹ������Һ��Ũ��ƫ����ʹ��������ʵ����ʵ���ƫС����ʹ��Һ�����ƫ��IJ���������ʹ������Һ��Ũ��ƫ�ݴ˷�����
��2���ܽ��ת��ʹ�ò�������
��3������n=c��V��m=n��M��������Ҫ��CuSO4?5H2O��������
��4��ͭ���ӳ���ɫ��ͭ���Ӽ��飺����Һ�м����������ƣ�����ɫ�������ɣ���������ӵļ��飺����Һ�м�������ʹ֮�ữ���ټ���BaCl2��Һ���а�ɫ����������
��5�����ʵ���Ũ����������������Ũ�ȹ�ʽC=
| n |
| V |
���
�⣺��1��ʵ����û��480mL������ƿ������Ҫ����500mL 1mol/L����Һ��Ӧѡ��500mL�������ƿ��
�ʴ�Ϊ��500��
��2���ܽ�ʱʹ�ò��������裻ת��ʱʹ�ò������������ʴ�Ϊ�����裻������
��3����Ҫ��CuSO4?5H2O������m=C��V��M=0.5L��1mol/L��250g/mol=125g���ʴ�Ϊ��125��
��4������ͭ��Һ����ɫ��ȡ������Һ���μ�����������Һ��������ɫ������֤����ͭ���ӣ���Ӧ���ӷ���ʽΪ��Cu2++2OH-=Cu��OH��2����
��ȡ������Һ���Թ��У��Ӽ���ϡ�����ữ���ټ��뼸���Ȼ�����Һ�����а�ɫ��������˵��������������ӣ����ӷ���ʽ��Ba2++SO42-=BaSO4����
�ʴ�Ϊ����������������Һ��Cu2++2OH-=Cu��OH��2����ϡ���ᡢ�Ȼ�����Һ��Ba2++SO42-=BaSO4����
��5������Һ���ƹ������ˮ���ݣ�����ƿʹ��ʱδ�����������ҺŨ����Ӱ�죬�ʴ�Ϊ����Ӱ�죻
�ڶ���ʱ�����۾����ӣ��ᵼ�¶���ʱ���������ˮƫ�٣�������Һ�����ƫС������Ũ�ȹ�ʽC=
��֪����õ���ҺŨ��ƫ�ߣ��ʴ�Ϊ��ƫ�ߣ�
�۶���ʱ��Һ�泬���̶��ߣ���������Һ���ƫ������Ũ�ȹ�ʽC=
��֪����õ���ҺŨ��ƫ�ͣ��ʴ�Ϊ��ƫ�ͣ�
�ʴ�Ϊ��500��
��2���ܽ�ʱʹ�ò��������裻ת��ʱʹ�ò������������ʴ�Ϊ�����裻������
��3����Ҫ��CuSO4?5H2O������m=C��V��M=0.5L��1mol/L��250g/mol=125g���ʴ�Ϊ��125��
��4������ͭ��Һ����ɫ��ȡ������Һ���μ�����������Һ��������ɫ������֤����ͭ���ӣ���Ӧ���ӷ���ʽΪ��Cu2++2OH-=Cu��OH��2����
��ȡ������Һ���Թ��У��Ӽ���ϡ�����ữ���ټ��뼸���Ȼ�����Һ�����а�ɫ��������˵��������������ӣ����ӷ���ʽ��Ba2++SO42-=BaSO4����
�ʴ�Ϊ����������������Һ��Cu2++2OH-=Cu��OH��2����ϡ���ᡢ�Ȼ�����Һ��Ba2++SO42-=BaSO4����
��5������Һ���ƹ������ˮ���ݣ�����ƿʹ��ʱδ�����������ҺŨ����Ӱ�죬�ʴ�Ϊ����Ӱ�죻
�ڶ���ʱ�����۾����ӣ��ᵼ�¶���ʱ���������ˮƫ�٣�������Һ�����ƫС������Ũ�ȹ�ʽC=
| n |
| V |
�۶���ʱ��Һ�泬���̶��ߣ���������Һ���ƫ������Ũ�ȹ�ʽC=
| n |
| V |
���������⿼����һ�����ʵ���Ũ����Һ�����ƣ���������ӵļ��飬�ѶȲ����������DZ�����ѵ����ڣ�ע��ѧ��������ʵ���Ũ�ȹ�ʽ�������жϵķ�����

��ϰ��ϵ�д�
�����Ŀ
��ѧ��Ƽ�����������������е���ϵ�������й�˵����ȷ���ǣ�������
| A��2009���ҹ�����ȼ��˰����ҵ�Ͻ�ʯ�ͷ���õ����ͣ���Ҫ�����˻�ѧ�仯 |
| B��2008��ŵ������ѧ�������о���ɫӫ�⵰�Ŀ�ѧ�ң����ֵ�������Һ����������Һ�����ö����ЧӦ |
C�� ���������������̷��¼�����Ⱦ��Ϊ�����谷���ṹ��ͼ������Ħ������Ϊl26g |
| D��2008��֧Ԯ�Ĵ�������������Ʒ��ʳ�ס����ʳ�ε���Ҫ��ѧ���ʷֱ������ᡢ��� |
�����и������ʰ����ʡ�������ᡢ��η���˳�����У�������ȷ���ǣ�������
| A�������ɱ������ᡢ�ռʳ�� |
| B����ơ��������ᡢ�ռʳ�� |
| C�������������������ᡢ�������� |
| D��ͭ������ͭ�����ᡢʯ��ˮ���Ȼ�ͭ |
ʵ����Ҫ��98%���ܶ�Ϊ1.84g?cm-3������������3.68mol/L������500mL��������3.68mol/L�����ᣬ������������ȷ�����в�����ʹ����������ҺŨ��ƫ�͵��ǣ�������
| A����ϡ�͵�����ת��������ƿ��δϴ���ձ��Ͳ����� |
| B�����ձ��ڵ�ϡ����������ƿ��ת��ʱ�����������ʹ����ϡ���ὦ��ƿ�� |
| C���ý�ͷ�ι�������ƿ�м�ˮʱ��Һ���������ƿ�̶��ߣ���ʱ�����ý�ͷ�ιܽ�ƿ�ڶ���Һ��������ʹ��Һ��Һ����̶������� |
| D���ý�ͷ�ι�������ƿ�м���ˮʱ�����ӹ۲���Һ��Һ��������ƿ�̶������� |
���н����͵�ˮ�еĵⵥ�ʷ��������ʵ�����˵����ȷ���ǣ�������
| A��Ӧ��ʹ����ȡ�ķ�������ѡ�þƾ�����ȡ�� |
| B����ȡʹ�õ���Ҫ�����Ƿ�Һ©������ʹ��ǰҪ�ȼ������Ƿ�©Һ |
| C�����÷ֲ��ⵥ��һ�������ϲ�Һ���У�Ӧ�ӷ�Һ©�����Ͽڵ��� |
| D���ӷ�Һ©���з�����ľ��Ǵ����ĵⵥ�� |
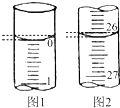 ijѧ��������֪���ʵ���Ũ�ȵ��������ⶨδ֪���ʵ���Ũ�ȵ�����������Һʱ��ѡ���̪��ָʾ��������д���пհף�
ijѧ��������֪���ʵ���Ũ�ȵ��������ⶨδ֪���ʵ���Ũ�ȵ�����������Һʱ��ѡ���̪��ָʾ��������д���пհף�