��Ŀ����
10������ͼ״�������ȼ�ϵ�أ����й����Ŀ�����Ӧ��ǰ�����ش���������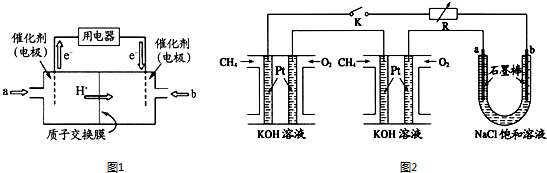
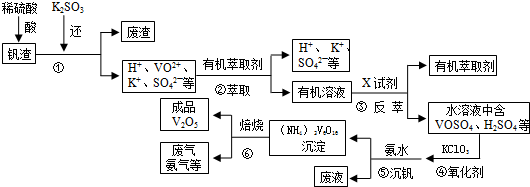
��1���״�ȼ�ϵ�أ����DMFC�����ڽṹ������ת���ʸߡ��Ի�������Ⱦ������Ϊ������Դ�����Ʒ��Խ��Խ�ܵ���ע��DMFC����ԭ����ͼ1��ʾ��ͨ��a����ĵ缫��ԭ��صĸ����������������������缫��ӦʽΪCH3OH+H2O-6e-�TCO2+6H+��
��2��ij�о�С�齫��������ȼ�ϵ�ش�������Ϊ��Դ�����б����Ȼ�����Һ���ʵ�飬��ͼ2��ʾU�ι����Ȼ�����Һ�����Ϊ800ml���պ�K����ÿ����ؼ���������Ϊ0.224L����������ҷ�Ӧ��ȫ����������ͨ�����صĵ���Ϊ7.72��103C�������ڳ���F=9.65��104C/mol����������������ȫ���ݳ���������Һ��Ͼ��ȣ�����U�ι�����Һ��pHΪ13��
���� ��1��ȼ�ϵ���У�ͨ��ȼ�ϵĵ缫Ϊ����������������Ӧ���ӳɱ��������ɶ�����̼��
��2������Ȼ�����Һ�Ļ�ѧ����ʽΪ2NaCl+2H2O$\frac{\underline{\;���\;}}{\;}$2NaOH+H2��+Cl2������֪��ϵʽ1 mol CH4��8 mol e-��8molNaOH���㣮
��� �⣺��1��ȼ�ϵ���У�ͨ��ȼ�ϵĵ缫Ϊ����������������Ӧ���ӳɱ��������ɶ�����̼���缫����ʽΪ��
�ʴ�Ϊ������CH3OH+H2O-6e -�TCO2+6H+��
��2������Ȼ�����Һ�Ļ�ѧ����ʽΪ2NaCl+2H2O$\frac{\underline{\;���\;}}{\;}$2NaOH+H2��+Cl2������֪��ϵʽ1 mol CH4��8 mol e-��8molNaOH������ÿ����ؼ���ͨ����Ϊ0.224L����״����������0.08molNaOH��c��NaOH��=$\frac{0.08mol}{0.8L}$=0.1mol/L��pH=13������ͨ���ĵ���Ϊ$\frac{0.224L}{22.4L/mol}$��8��9.65��104C•mol-1=7.72��103C��������Ȼ������ȼ�ϵ�أ������ӵĴ�����ֻ����һ���صļ��������㣩��
�ʴ�Ϊ��7.72��103C��13��
���� �����ۺϿ���绯ѧ֪ʶ���漰�缫�ж���缫��Ӧʽ��д�����⣬����ʱע���������ԭ�ĽǶ��ж�ԭ��ص��������Լ��缫����ʽ����д���������ѵ���״���Ϊ�缫����ʽ����д����Ŀ�Ѷ��еȣ�

| A�� | $\frac{ab}{5}$��100% | B�� | $\frac{2ab}{5}$��100% | C�� | $\frac{2ab}{5b}$��100% | D�� | $\frac{ab}{5a}$��100% |
| A�� | pH=4.3��CH3COOH��CH3COONa�����Һ�У�c��Na+����c��CH3COO-�� | |
| B�� | Ũ��Ϊ0.2 mol/L��CH3COOH��Һ��Ũ��Ϊ0.1mol/L��NaOH��Һ�������Ϻ�c��CH3COO -��-c��CH3COOH��=2[c��H+��-c��OH-��] | |
| C�� | ����Ũ��Һ������ˮϡ�ͣ�$\frac{c��C{H}_{3}COOH��}{{c}^{2}��{H}^{+}��}$�������� | |
| D�� | �������ʵ�����NaHC2O4��Na2C2O4����Һ��3c��Na+��=2[c��HC2O4-��+c��C2O42-��+c��H2C2O4��] |
| X | Y | ||
| Z | M | R |
| A�� | Ԫ�صķǽ����Դ���Ϊ��Y��X��M | |
| B�� | ��̬�⻯���ȶ��ԣ�M��R | |
| C�� | Z����������������ά | |
| D�� | ����������Ӧˮ��������ԣ�Y��X |
| A�� | ԭ�Ӱ뾶��E��B��C��D | |
| B�� | A�ֱ���B��C��D��E�γɵļ������У��ȶ�����õĺͷе���ߵĶ���AD | |
| C�� | E���������ȶ�������Ԫ������Ȼ��ֻ���Ի���̬��ʽ���� | |
| D�� | ����������һ�����й��ۼ������ܺ������Ӽ� |
| A�� | �ƾ� | B�� | H2 | C�� | Na2SO4 | D�� | SO2 |
| A�� | CH3-CH2-NO2��H2N-CH2-COOH��Ϊͬ���칹�� | |
| B�� | H��D��T��Ϊͬλ�� | |
| C�� | O2��O3��Ϊͬ�������� | |
| D�� | CH3CH2CH2CH3�� ������ȫ��ͬ ������ȫ��ͬ |