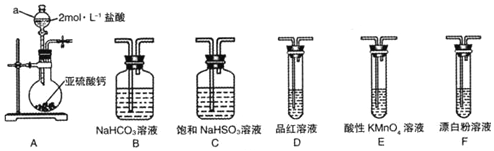
��Ŀ����
12��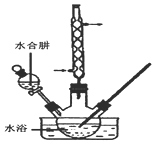 �⻯����һ�ְ�ɫ�ᾧ��ĩ��ҽ���Ͽ����ڼ�״���������μ�����̵���ȣ�ʵ������NaOH�����ʵ��ˮ����Ϊԭ���Ƶã�����װ����ͼ��ʾ��
�⻯����һ�ְ�ɫ�ᾧ��ĩ��ҽ���Ͽ����ڼ�״���������μ�����̵���ȣ�ʵ������NaOH�����ʵ��ˮ����Ϊԭ���Ƶã�����װ����ͼ��ʾ��ʵ�鲽�����£�
�������������ƿ�м���8.2gNaOH��30mLˮ��������ȴ��
��������Ƶõ�NaOH��Һ�м���25.4g���ʵ⣬��������������������60〜70������Ӧ��֣�
����������������Һ�м����Թ�����N2H4•H2O��ˮ���£�����ԭNaIO��NaI33����Nal��Һ��Ʒ�Ϳ����г�����ij���壮
���������Һ���м���1.0g����̿����а�Сʱ��Ȼ����Һ�����̿���룮
���������Ũ�����ᾧ�����˼�����ò�Ʒ24.3g��
��1��������¶Ȳ��˳���70���ԭ���Ƿ�ֹ���������ò��跴Ӧ��ȫ�������������������Һ�ӽ���ɫ��
��2�������N2H4•H2O��ԭNalO3�Ļ�ѧ����ʽΪ3N2H4•H2O+2NaIO3=2NaI+3N2��+9H2O��
��3������ܼ������̿��Ŀ������ɫ��������Һ�����̿���롱�ķ����dz��ȹ��ˣ�
��4������ʵ���Ʒ��Ϊ81%��
���� �����������ƣ�����������Ʒ�����Ӧ��3I2+6NaOH=5NaI+NaIO3+3H2O������ˮ���µõ�������NaI����������Ӧ��3N2H4•H2O+2NaIO3=2NaI+3N2��+9H2O���õ���NaI��Һ������Ũ������ȴ�ᾧ�ɵõ�NaI��
��1���¶ȳ���70��ⵥ����������������еⵥ������NaI��NaIO3����Ӧ��ȫʱ����Ϊ�����������Һ�ӽ���ɫ��
��2�������N2H4•H2O��ԭNalO3�Ļ�ѧ����ʽΪ3N2H4•H2O+2NaIO3=2NaI+3N2��+9H2O��
��3�����ݻ���̿�������Է�����ͨ�����ȹ��˽�����̿��⻯����Һ���룻
��4��8.2gNaOH��25.4g���ʵⷴӦ���������ƹ������ⵥ�ʷ�Ӧ��ȫ���ɵⵥ�ʼ������ɵ�NaI��NaIO3������NaIO3������N2H4•H2O��Ӧ���õ�NaI���ɴ˼���õ��������ɵ�NaI���ټ�����ʿɵã�
��� �⣺��1���ⵥ����������������¶Ȳ��˳���70�棻������еⵥ������NaI��NaIO3����Ӧ��ȫʱ����Ϊ�����������Һ�ӽ���ɫ��
�ʴ�Ϊ����ֹ�������������������Һ�ӽ���ɫ��
��2�������N2H4•H2O��ԭNalO3�Ļ�ѧ����ʽΪ3N2H4•H2O+2NaIO3=2NaI+3N2��+9H2O��
�ʴ�Ϊ��3N2H4•H2O+2NaIO3=2NaI+3N2��+9H2O��
��3������̿���������ԣ�����ɫ��ͨ�����ȹ��˽�����̿��⻯����Һ���룻
�ʴ�Ϊ����ɫ�����ȹ��ˣ�
��4��8.2gNaOH��25.4g���ʵⷴӦ���������ƹ������ⵥ�ʷ�Ӧ��ȫ������������Ʒ�����Ӧ��3I2+6NaOH=5NaI+NaIO3+3H2O�������ɵ�NaI������Ϊ��$\frac{25.4��750}{762}$=25g�����ɵ�NaIO3��N2H4•H2O��Ӧ���õ�NaI����ӦΪ3N2H4•H2O+2NaIO3=2NaI+3N2��+9H2O����6I2��2NaIO3��2NaI���ò����ɵ�NaI����Ϊ��$\frac{25.4��300}{1524}$=5g�������������ɵ�NaIΪ25g+5g=30g��ʵ���Ʒ��Ϊ$\frac{24.3}{30}��100%$=81%��
�ʴ�Ϊ����81%��
���� ���⿼���������Ʊ�������ƣ���Ҫ�����˻�ѧ����ʽ����д��ʵ�鷽�����ۡ�������ԭ��Ӧ�����ʼ��㣬��Ŀ�Ѷ��еȣ������ڿ���ѧ���ķ�������ͽ�������������

| A�� | H2 | B�� | CO | C�� | CH4 | D�� | C2H4 |
| A�� | �þƾ���������������������ۻ��������䣬˵��Al2O3��Al | |
| B�� | ��¯ˮ��CaSO4����Na2CO3��Һ���ݣ��������ܽ�ȥ����˵��Ksp��CaCO3��CaSO4 | |
| C�� | ��Na2SiO3��Һ��ͨ������CO2���壬�а�ɫ�������ɣ�˵�����ԣ�H2CO3��H2SiO3 | |
| D�� | ������ͨ��������Һʱ������һ�������ġ�ͨ·����˵����ɢ������ֱ����1-100nm֮�� |
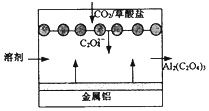
| A�� | ��װ���ǽ�����ת��Ϊ��ѧ�� | |
| B�� | �����ĵ缫��ӦΪ��C2O42--2e-=2CO2 | |
| C�� | ÿ�õ�1 mol����������·��ת��3 mol���� | |
| D�� | ���øü����ɲ������е�CO2 |
| A�� | ϡ��Ũ����ʱ����ˮ����������ע������ | |
| B�� | ��ȼ����ǰ���ȼ��������Ĵ��� | |
| C�� | ����Һ��ʱ���Թܿڲ������� | |
| D�� | ���ö����ЧӦ���������Һ |
| A�� | 2NaBr+Cl2�TBr2+2NaCl | |
| B�� | AlCl3+3NaAlO2+6H2O�T4Al��OH��3��+3NaCl | |
| C�� | 2H2S+SO2�T3S��+2H2O | |
| D�� | Cl2+H2O�THCl+HClO |
| A�� | Ũ��ˮ�еμ�FeCl3������Һ���Ƶ�Fe��OH��3���� | |
| B�� | ��ըʳ��Ļ����ͺ�ţ�Ͷ��ǿ������ı������� | |
| C�� | �������֡�ˮ��������ˮ��Ϊ����� | |
| D�� | KClO3��SO3����ˮ���ܵ��磬��KClO3��SO3Ϊ����� |
| A�� | NH4+��K+��CO32-��Cl- | B�� | Al3+��Cu2+��S2-��Cl- | ||
| C�� | NH4+��Cu2+��Cl-��NO3- | D�� | Fe3+��K+��SCN-��NO3- |