��Ŀ����
5��ijͬѧ��15mol•L-1Ũ���ᣬ�����в�����2505mL0.1mol•L-1H2SO4��Һ����ش��й����⣮| ʵ�鲽�� | �й����� |
| ��1����������Ũ�������� | ��ȡŨ���ᣨ1.7��mL |
| ��2���� ȡŨ���� | ��Ҫ�õ�����Ҫ�����ǣ�5mL��Ͳ |
| ��3���� Ũ�������Ų���������ע��ʢ��ˮ���ձ��в����� | �ò���Ҫ����������ϸ���ž��¹ʷ��� |
| ��4������Һ��ȴ�����£����ձ�����Һת����250mL����ƿ�� | Ҫע���ϴ��Һһ��ת��������ƿ�� |
| ��5��������ƿ�м�����ˮ���̶��� | �ڽ��д˲���ʱ����ˮ���̶���1-2cm�������ȷ�����ǣ����ý�ͷ�ι���εμӵ���Һ����ʹ���̶������� |
a�������� b���ձ� c����ƽ d��ͷ�ι�
��2���ڽ��Тܲ�����ʱ����������Һ��������ƿ�⣬��������Һ��Ũ�Ƚ�ƫ�ͣ���дƫ�ߡ�ƫ�͡���Ӱ�죮��ͬ����������ʱ����ƿû�и����������ҺŨ�Ƚ���Ӱ�죮
��3��ij�о���ԱӦ����ͼ�о����ʵ����ʣ���������A����Ҫ�ɷ��������������ǿ�����ˮ��������ش��������⣺
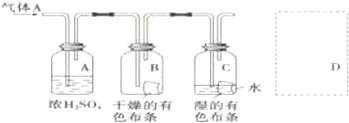
��Ũ����������Ǹ������
��ͨ��Cl2�Ӽ���ƿB�и������ɫ����������ɵó��Ľ����Ǹ�������������Ư���ԴӼ���ƿC��ʪ�����ɫ����������ɵó��Ľ����Ǵ��������Ư���ԣ�
�۴��������ʵķ���������������ʵ����ƻ������¹�����������ͼD����ͼ����ʽ�����˷��¹������Ĵ�ʩ��
���� ����������Һ���ѡ����ʵ�����ƿ��������Һϡ���������ʵ����ʵ������������ҪŨ�����������ȡŨ�����õ�����Ҫ����Ϊ��Ͳ������Ũ�������ѡ����ʹ�����Ͳ��
ע��Ũ����ϡ�͵���ȷ������ע�ⶨ�ݵ���ȷ������
��1����������һ�����ʵ���Ũ����Һ��������ѡ����Ҫ�������ж��ò�����������
��2���������������ʵ����ʵ�������Һ�����Ӱ�죬����C=$\frac{n}{V}$������������
��3�������Ȼ��⡢ˮ������������ͨ��Ũ������и����ȥˮ����������B��C�У�����BC�в�����ɫ�ı仯�ó����ۣ�����������Ư���ԣ�����Ư���Ե��Ǵ����ᣬע�������ж���Ҫ����β�������������ܹ���Ӧ��������ʢ���������Ƶ��ձ����չ�����������
��� �⣺��250mL0.1mol•L-1H2SO4��Һ��Ӧѡ��250mL����ƿ������Ҫ15mol•L-1Ũ�������ΪV����������Һϡ���������ʵ����ʵ�������ã�V��15mol/L=250mL��0.1mol/L�����V=1.7mL����Ҫ�õ�����Ҫ����Ϊ5mL��Ͳ��Ũ����ϡ�͵���ȷ��������Ũ�������Ų���������ע��ʢ��ˮ���ձ��в����裻
���ݵ���ȷ����Ϊ��������ƿ�м�ˮ���̶���1-2cm������ý�ͷ�ι���εμӵ���Һ����ʹ���̶������У�
�ʴ�Ϊ��
| ʵ�鲽�� | �й����� |
| ��1����������Ũ�������� | ��ȡŨ���ᣨ 1.7��mL |
| ��2���� ȡŨ���� | ��Ҫ�õ�����Ҫ�����ǣ�5mL��Ͳ |
| ��3���� Ũ���� ���Ų���������ע��ʢ��ˮ���ձ��в����� | �ò���Ҫ����������ϸ���ž��¹ʷ��� |
| ��4������Һ��ȴ�����£����ձ�����Һת���� 250mL����ƿ�� | Ҫע���ϴ��Һһ��ת��������ƿ�� |
| ��5��������ƿ�м�����ˮ���̶��� | �ڽ��д˲���ʱ����ˮ���̶���1-2cm�������ȷ�����ǣ����ý�ͷ�ι���εμӵ���Һ����ʹ���̶������� |
��ѡ��c��
��2���ڽ��Тܲ�����ʱ����������Һ��������ƿ�⣬����������ģ����ʵ����ʵ���ƫС����������Һ��Ũ�Ƚ�ƫ�ͣ�
������ʱ����ƿû�и�������ʵ����ʵ�������Һ������������Ӱ�죬��������ҺŨ�Ƚ�����Ӱ�죻
�ʴ�Ϊ��ƫ�ͣ���Ӱ�죻
��3����Ũ���������ˮ�ԣ��ܹ�����������
�ʴ�Ϊ������������
��ͨ��Cl2�Ӽ���ƿB�и������ɫ��������ɫ��֪����������Ư���ԣ��ӴӼ���ƿC��ʪ�����ɫ����������ɵó��Ľ�����������ˮ��Ӧ���ɵĴ��������Ư���ԣ�
�ʴ�Ϊ����������������Ư���ԣ����������Ư���ԣ�
�������ж���Ӧ����β�����������������ŷŵ������У����ü���Һ�����գ�����ʽΪCl2+2NaOH=NaCl+NaClO+H2O��װ��ͼΪ
 ��
���ʴ�Ϊ��
 ��
������ ���⿼����һ�����ʵ���Ũ����Һ�����ƣ�����������̽��ʵ�飬��ȷ����ԭ���Ͳ����ǽ���ؼ���ע��̽��ʵ����Ƶ������ԣ���Ŀ�ѶȲ���

| A�� | ������Ҫ���ȵķ�Ӧ�������ȷ�Ӧ | |
| B�� | ���н���Ԫ���γɵĻ����ﶼ�����ӻ����� | |
| C�� | ��������Ԫ�ص�������������ԭ��������ϵĵ����� | |
| D�� | ���й��ۻ����������ÿ��ԭ������㶼�ﵽϡ��������ȶ��ṹ |
| A�� | ԭ��ؽ���ѧ��ת��Ϊ���� | |
| B�� | ԭ��ؽ�����ת��Ϊ��ѧ�� | |
| C�� | �кͷ�Ӧ����˵���кͷ�Ӧǰ���������غ� | |
| D�� | ���ȷ�Ӧ������������������ڷ�Ӧ��������� |
| A�� | H2XO3 | B�� | HXO3 | C�� | HXO | D�� | H2XO4 |
| ���ۼ� | H-H | F-F | H-F | H-Cl | H-I |
| E��kJ•mol-1 �� | 436 | 157 | 568 | 432 | 298 |
| A�� | 432 kJ•mol -1��E��H-Br����298 kJ•mol -1 | |
| B�� | �������ȶ��Ĺ��ۼ���H-F�� | |
| C�� | H 2 ��g����2H ��g����H=+436 kJ•mol -1 | |
| D�� | H 2 ��g��+F 2 ��g��=2HF��g����H=-25 kJ•mol -1 |
| A�� | Ca2CO3+2HCl�TCaCl2+H2O+CO2�� | B�� | 2H2O2$\frac{\underline{\;MnO_2\;}}{\;}$ 2H2O+O2�� | ||
| C�� | Cu+4HNO3��ϡ���TCu��NO3��2+2NO��+2H2O | D�� | NaCl+AgNO3�TAgCl��+NaNO3 |
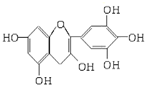 �����ҹ��Ĵ�ͳ��Ʒ����Ҷ�к��еIJ��ӿ������ʳƷ���Ӽ��ж������к��ĺϳɿ������������ڶ���ʳƷ���ʵȣ���ͼ��ʾ�Dz����к�����ߵ�һ�ֶ�����A�Ľṹ��ʽ���������ֶ�����A���й�������ȷ���ǣ�������
�����ҹ��Ĵ�ͳ��Ʒ����Ҷ�к��еIJ��ӿ������ʳƷ���Ӽ��ж������к��ĺϳɿ������������ڶ���ʳƷ���ʵȣ���ͼ��ʾ�Dz����к�����ߵ�һ�ֶ�����A�Ľṹ��ʽ���������ֶ�����A���й�������ȷ���ǣ�������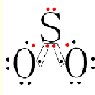 ��
��