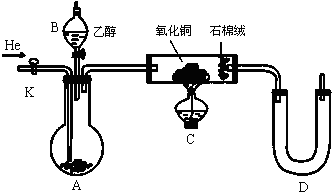
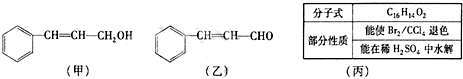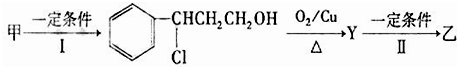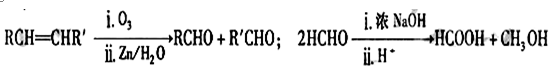��Ŀ����
Ϊ̽����Ȳ����ļӳɷ�Ӧ����ͬѧ��Ʋ�����������ʵ�飺��ȡһ������ҵ�õ�ʯ��ˮ��Ӧ�������ɵ�����ͨ����ˮ�У�������Һ��ɫ����֤����Ȳ����ˮ�����˼ӳɷ�Ӧ��
��ͬѧ�����ڼ�ͬѧ��ʵ���У���ɫ�����Һ������������ɫ���ǣ��Ʋ����Ƶõ���Ȳ�л����ܺ���������ԭ�Ե��������壬�ɴ�����������ȳ�ȥ֮��������ˮ��Ӧ��
����ش��������⣺
��1��д��̼���Ƶĵ���ʽ ����ͬѧʵ����������Ҫ�Ļ�ѧ����ʽΪ �� ��
��2��Ϊ�˼�����ʯ��ˮ�ķ�Ӧ���ʣ�ʵ��ʱ���� ����ˮ��
��3����ͬѧ��Ƶ�ʵ�� �����ܻ��ܣ���֤��Ȳ���巢���ӳɷ�Ӧ���������� ��
��a��ʹ��ˮ��ɫ�ķ�Ӧ��δ���Ǽӳɷ�Ӧ
��b��ʹ��ˮ��ɫ�ķ�Ӧ�����Ǽӳɷ�Ӧ
��c��ʹ��ˮ��ɫ�����ʣ�δ������Ȳ
��d��ʹ��ˮ��ɫ�����ʣ�������Ȳ
��4����ͬѧ�Ʋ����Ȳ�бض�����һ���������壬������ˮ��Ӧ�Ļ�ѧ����ʽ�� ������֤�������� ��ȥ��
��5��Ϊ��֤��Ȳ����ˮ�ķ�Ӧ�Ǽӳɷ�Ӧ������ȡ����Ӧ����ͬѧ�������pH��ֽ�����Է�Ӧ�����Һ�����ԣ������� ��
��ͬѧ�����ڼ�ͬѧ��ʵ���У���ɫ�����Һ������������ɫ���ǣ��Ʋ����Ƶõ���Ȳ�л����ܺ���������ԭ�Ե��������壬�ɴ�����������ȳ�ȥ֮��������ˮ��Ӧ��
����ش��������⣺
��1��д��̼���Ƶĵ���ʽ
��2��Ϊ�˼�����ʯ��ˮ�ķ�Ӧ���ʣ�ʵ��ʱ����
��3����ͬѧ��Ƶ�ʵ��
��a��ʹ��ˮ��ɫ�ķ�Ӧ��δ���Ǽӳɷ�Ӧ
��b��ʹ��ˮ��ɫ�ķ�Ӧ�����Ǽӳɷ�Ӧ
��c��ʹ��ˮ��ɫ�����ʣ�δ������Ȳ
��d��ʹ��ˮ��ɫ�����ʣ�������Ȳ
��4����ͬѧ�Ʋ����Ȳ�бض�����һ���������壬������ˮ��Ӧ�Ļ�ѧ����ʽ��
��5��Ϊ��֤��Ȳ����ˮ�ķ�Ӧ�Ǽӳɷ�Ӧ������ȡ����Ӧ����ͬѧ�������pH��ֽ�����Է�Ӧ�����Һ�����ԣ�������
���㣺����ʵ�鷽�������
ר�⣺ʵ�������
��������1������̼����Ϊ���ӻ�������д����ʽ��̼��������ˮ��Ӧ������Ȳ����Ȳ���в����ͼ���������ˮ�����ӳɷ�Ӧ��ʹ��ˮ��ɫ��
��2��Ϊ�˼�����ʯ��ˮ�ķ�Ӧ���ʣ�ʵ��ʱ���ñ���ʳ��ˮ����ˮ��
��3�����ɵ���Ȳ�л����������壬������ˮ����������ԭ��Ӧ��ʹ��ˮ��ɫ��
��4����ʯ�к��������ˮ��Ӧ�����������壬������л�ԭ�Կ��Ա���ˮ������������ͭ���������������⣻
��5������ȡ����Ӧ�������ǣ���ȥ�л����ش�
��2��Ϊ�˼�����ʯ��ˮ�ķ�Ӧ���ʣ�ʵ��ʱ���ñ���ʳ��ˮ����ˮ��
��3�����ɵ���Ȳ�л����������壬������ˮ����������ԭ��Ӧ��ʹ��ˮ��ɫ��
��4����ʯ�к��������ˮ��Ӧ�����������壬������л�ԭ�Կ��Ա���ˮ������������ͭ���������������⣻
��5������ȡ����Ӧ�������ǣ���ȥ�л����ش�
���
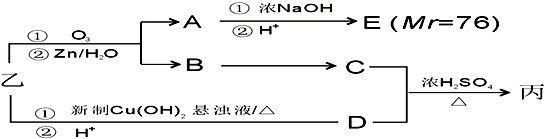
�⣺��1��̼����Ϊ���ӻ�����ʵ���ʽΪ��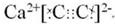 ��̼��������ˮ��Ӧ������Ȳ��ˮ����Ӧ����ʽΪCaC2+2H2O��Ca��OH��2+C2H2������Ȳ���в����ͼ���������ˮ�����ӳɷ�Ӧ��ʹ��ˮ��ɫ����Ӧ����ʽΪCH��CH+2Br2��CHBr2-CHBr2��
��̼��������ˮ��Ӧ������Ȳ��ˮ����Ӧ����ʽΪCaC2+2H2O��Ca��OH��2+C2H2������Ȳ���в����ͼ���������ˮ�����ӳɷ�Ӧ��ʹ��ˮ��ɫ����Ӧ����ʽΪCH��CH+2Br2��CHBr2-CHBr2��
�ʴ�Ϊ��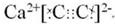 ��CaC2+2H2O�TCa��OH��2+C2H2����CH��CH+2Br2��CHBr2-CHBr2��
��CaC2+2H2O�TCa��OH��2+C2H2����CH��CH+2Br2��CHBr2-CHBr2��
��2��Ϊ�˼�����ʯ��ˮ�ķ�Ӧ���ʣ�ʵ��ʱ���ñ���ʳ��ˮ����ˮ���ʴ�Ϊ������ʳ��ˮ��
��3�����ɵ���Ȳ�л����������壬������ˮ����������ԭ��Ӧ��ʹ��ˮ��ɫ���ʴ�Ϊ�����ܣ�ac��
��4����ʯ�к��������ˮ��Ӧ�����������壬����������л�ԭ�ԣ�������ˮ����������ԭ��Ӧ��������廯�⣬��Ӧ����ʽΪBr2+H2S�TS��+2HBr������ͭ����������������ͭ����������������ͭ�������������壻
�ʴ�Ϊ��Br2+H2S�TS��+2HBr��CuSO4��Һ��
��5��Ϊ��֤��һ��Ӧ�Ǽӳɶ�����ȡ������ͬѧ�������pH��ֽ�����Է�Ӧ����Һ�����ԣ���������ȡ����Ӧ���ض�����HBr����Һ���Խ���������ǿ���ʿ���pH��ֽ��֤��
�ʴ�Ϊ����������ȡ����Ӧ���ض�����HBr����Һ������Խ���������ǿ�����ӳɷ�Ӧ��������HBr���ʿ���PH��ֽ���飮
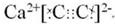 ��̼��������ˮ��Ӧ������Ȳ��ˮ����Ӧ����ʽΪCaC2+2H2O��Ca��OH��2+C2H2������Ȳ���в����ͼ���������ˮ�����ӳɷ�Ӧ��ʹ��ˮ��ɫ����Ӧ����ʽΪCH��CH+2Br2��CHBr2-CHBr2��
��̼��������ˮ��Ӧ������Ȳ��ˮ����Ӧ����ʽΪCaC2+2H2O��Ca��OH��2+C2H2������Ȳ���в����ͼ���������ˮ�����ӳɷ�Ӧ��ʹ��ˮ��ɫ����Ӧ����ʽΪCH��CH+2Br2��CHBr2-CHBr2���ʴ�Ϊ��
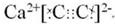 ��CaC2+2H2O�TCa��OH��2+C2H2����CH��CH+2Br2��CHBr2-CHBr2��
��CaC2+2H2O�TCa��OH��2+C2H2����CH��CH+2Br2��CHBr2-CHBr2����2��Ϊ�˼�����ʯ��ˮ�ķ�Ӧ���ʣ�ʵ��ʱ���ñ���ʳ��ˮ����ˮ���ʴ�Ϊ������ʳ��ˮ��
��3�����ɵ���Ȳ�л����������壬������ˮ����������ԭ��Ӧ��ʹ��ˮ��ɫ���ʴ�Ϊ�����ܣ�ac��
��4����ʯ�к��������ˮ��Ӧ�����������壬����������л�ԭ�ԣ�������ˮ����������ԭ��Ӧ��������廯�⣬��Ӧ����ʽΪBr2+H2S�TS��+2HBr������ͭ����������������ͭ����������������ͭ�������������壻
�ʴ�Ϊ��Br2+H2S�TS��+2HBr��CuSO4��Һ��
��5��Ϊ��֤��һ��Ӧ�Ǽӳɶ�����ȡ������ͬѧ�������pH��ֽ�����Է�Ӧ����Һ�����ԣ���������ȡ����Ӧ���ض�����HBr����Һ���Խ���������ǿ���ʿ���pH��ֽ��֤��
�ʴ�Ϊ����������ȡ����Ӧ���ض�����HBr����Һ������Խ���������ǿ�����ӳɷ�Ӧ��������HBr���ʿ���PH��ֽ���飮
���������⿼������Ȳ���Ʊ�������ʵ�飬��Ŀ�ѶȲ�������ע����Ȳ�ij��Ӻ�����ʵ�飬ע������ˮ��Ӧ�����ʣ�

��ϰ��ϵ�д�
�����Ŀ
�����ʷ����У�ǰ�߰������ߵ��ǣ�������
| A������������� |
| B����Һ������ |
| C����ɢϵ����Һ |
| D����������������� |
 ������A�����ϣ��������������֪����Է�������Ϊ136������ʽΪC8H8O2��A�ĺ˴Ź���������4���������֮��Ϊ1��2��2��3��A������ֻ��һ�������ұ�����ֻ��һ��ȡ����������������˴Ź���������ͼ������A������˵���У���ȷ���ǣ�˫ѡ����������
������A�����ϣ��������������֪����Է�������Ϊ136������ʽΪC8H8O2��A�ĺ˴Ź���������4���������֮��Ϊ1��2��2��3��A������ֻ��һ�������ұ�����ֻ��һ��ȡ����������������˴Ź���������ͼ������A������˵���У���ȷ���ǣ�˫ѡ����������| A��A����������������һ���������ܷ���ˮ�ⷴӦ |
| B��A��һ�������¿���4 mol H2�����ӳɷ�Ӧ |
| C����������A���ӽṹ�������л���ֻ��1�� |
| D����A����ͬ������ͬ���칹��ֻ��2�� |
����˵����ȷ���ǣ�������
| A��ϡH2SO4�ܵ��磬����ϡH2SO4�ǵ���� |
| B��ʧȥ���Ӷ�Ļ�ԭ������ԭ��һ��ǿ |
| C�������Ƿ��ж����ЧӦ����ɢϵ��Ϊ��Һ���������Һ |
| D�����ݽ�����෨MgSO4����þ������������ |