��Ŀ����
ҩ�│�������ĺϳ�ԭ��F�����̲��Ͼ�̼���������PC���ĺϳ�·������ͼ��ʾ��

��֪��
��
�����뺬�ǻ��Ļ�����ɷ���������������Ӧ��RCOOR��+R��OH
RCOOR��+R��OH
��R��R�䡢R�����������
��ش�
��1��C�ɱ�ϩ����Ӧ�١��ۺϳɣ�C �ĺ˴Ź�������ֻ��һ�ַ壮
a���ٵķ�Ӧ������ ��
b���ڵķ�Ӧ�Լ��������� ��
c���۵Ļ�ѧ����ʽ�� ��
��2��9.4g��D�뱥����ˮ��ȫ��Ӧ����33.1g��ɫ������D�Ľṹ��ʽ�� ��
��3��C��D��Ӧ����˫��A�Ļ�ѧ����ʽ�� ��
��4��F�ж���ͬ���칹�壬������������������ͬ���칹��Ľṹ��ʽ�� ��
�ٺ��м� �ں���̼̼˫�� ���ܷ���������Ӧ ��������
��5��E�Ľṹ��ʽ�� ��

��֪��
��

�����뺬�ǻ��Ļ�����ɷ���������������Ӧ��RCOOR��+R��OH
| ���� |
��R��R�䡢R�����������
��ش�
��1��C�ɱ�ϩ����Ӧ�١��ۺϳɣ�C �ĺ˴Ź�������ֻ��һ�ַ壮
a���ٵķ�Ӧ������
b���ڵķ�Ӧ�Լ���������
c���۵Ļ�ѧ����ʽ��
��2��9.4g��D�뱥����ˮ��ȫ��Ӧ����33.1g��ɫ������D�Ľṹ��ʽ��
��3��C��D��Ӧ����˫��A�Ļ�ѧ����ʽ��
��4��F�ж���ͬ���칹�壬������������������ͬ���칹��Ľṹ��ʽ��
�ٺ��м� �ں���̼̼˫�� ���ܷ���������Ӧ ��������
��5��E�Ľṹ��ʽ��
���㣺�л���ĺϳ�
ר�⣺
��������ϩ����ϵ�з�Ӧ�õ�C��C��D��Ӧ�õ�˫��A��C�ܺ������ᷢ����Ϣ�еļӳɷ�Ӧ�����������Ϣ��˫��A�Ľṹ��֪��C�� ����B��
����B�� ��D��
��D�� ��A��
��A��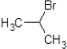 ����ͪ��HCN��Ӧ���ữ�õ���CH3��2C��OH��COOH��M����Է���������86���ȣ�CH3��2C��OH��COOHС18��Ӧ�ǣ�CH3��2C��OH��COOH��Ũ���ᡢ���������·�����������ȥ1����ˮ����F����F����ΪCH2=C��CH3��COOH��
����ͪ��HCN��Ӧ���ữ�õ���CH3��2C��OH��COOH��M����Է���������86���ȣ�CH3��2C��OH��COOHС18��Ӧ�ǣ�CH3��2C��OH��COOH��Ũ���ᡢ���������·�����������ȥ1����ˮ����F����F����ΪCH2=C��CH3��COOH��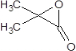 ��̼���������D��
��̼���������D�� ����Ӧ�õ�E��״�����EΪ
����Ӧ�õ�E��״�����EΪ ��˫��A��E��Ӧ�õ�������PC����PCΪ
��˫��A��E��Ӧ�õ�������PC����PCΪ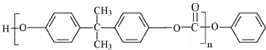 ��
��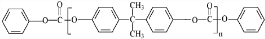 ���ݴ˽��
���ݴ˽��
 ����B��
����B�� ��D��
��D�� ��A��
��A�� ����ͪ��HCN��Ӧ���ữ�õ���CH3��2C��OH��COOH��M����Է���������86���ȣ�CH3��2C��OH��COOHС18��Ӧ�ǣ�CH3��2C��OH��COOH��Ũ���ᡢ���������·�����������ȥ1����ˮ����F����F����ΪCH2=C��CH3��COOH��
����ͪ��HCN��Ӧ���ữ�õ���CH3��2C��OH��COOH��M����Է���������86���ȣ�CH3��2C��OH��COOHС18��Ӧ�ǣ�CH3��2C��OH��COOH��Ũ���ᡢ���������·�����������ȥ1����ˮ����F����F����ΪCH2=C��CH3��COOH�� ��̼���������D��
��̼���������D�� ����Ӧ�õ�E��״�����EΪ
����Ӧ�õ�E��״�����EΪ ��˫��A��E��Ӧ�õ�������PC����PCΪ
��˫��A��E��Ӧ�õ�������PC����PCΪ ��
�� ���ݴ˽��
���ݴ˽�����
�⣺��ϩ����ϵ�з�Ӧ�õ�C��C��D��Ӧ�õ�˫��A��C�ܺ������ᷢ����Ϣ�еļӳɷ�Ӧ�����������Ϣ��˫��A�Ľṹ��֪��C�� ����B��
����B�� ��D��
��D�� ��A��
��A�� ����ͪ��HCN��Ӧ���ữ�õ���CH3��2C��OH��COOH��M����Է���������86���ȣ�CH3��2C��OH��COOHС18��Ӧ�ǣ�CH3��2C��OH��COOH��Ũ���ᡢ���������·�����������ȥ1����ˮ����F����F����ΪCH2=C��CH3��COOH��
����ͪ��HCN��Ӧ���ữ�õ���CH3��2C��OH��COOH��M����Է���������86���ȣ�CH3��2C��OH��COOHС18��Ӧ�ǣ�CH3��2C��OH��COOH��Ũ���ᡢ���������·�����������ȥ1����ˮ����F����F����ΪCH2=C��CH3��COOH�� ��̼���������D��
��̼���������D�� ����Ӧ�õ�E��״�����EΪ
����Ӧ�õ�E��״�����EΪ ��˫��A��E��Ӧ�õ�������PC����PCΪ
��˫��A��E��Ӧ�õ�������PC����PCΪ ��
�� ��
��
��1��a����Ӧ���DZ�ϩ��HBr�����ļӳɷ�Ӧ���ʴ�Ϊ���ӳɷ�Ӧ��
b����Ӧ����±����������ˮ�ⷴӦ����Ӧ�Լ��������ǣ���������ˮ��Һ�����ȣ��ʴ�Ϊ����������ˮ��Һ�����ȣ�
c����Ӧ�۵Ļ�ѧ����ʽ�ǣ�2CH3CH��OH��CH3+O2
2 +2H2O���ʴ�Ϊ��2CH3CH��OH��CH3+O2
+2H2O���ʴ�Ϊ��2CH3CH��OH��CH3+O2
2 +2H2O��
+2H2O��
��2��ͨ�����Ϸ���֪��D�DZ��ӣ��ṹ��ʽ�� ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��3��C��D��Ӧ����˫��A�Ļ�ѧ����ʽ�� ��
��
�ʴ�Ϊ�� ��
��
��4��F���ж���ͬ���칹�壬��������������F������ͬ���칹�壺�ٺ��м� �ں���̼̼˫�� ���ܷ���������Ӧ ���������࣬Ӧ�Ǽ����γɵ���������������ͬ���칹���У�HCOOCH=CHCH3��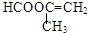 ���ʴ�Ϊ��HCOOCH=CHCH3��
���ʴ�Ϊ��HCOOCH=CHCH3��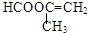 ��
��
��5��������������֪��E�Ľṹ��ʽ�� ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
 ����B��
����B�� ��D��
��D�� ��A��
��A�� ����ͪ��HCN��Ӧ���ữ�õ���CH3��2C��OH��COOH��M����Է���������86���ȣ�CH3��2C��OH��COOHС18��Ӧ�ǣ�CH3��2C��OH��COOH��Ũ���ᡢ���������·�����������ȥ1����ˮ����F����F����ΪCH2=C��CH3��COOH��
����ͪ��HCN��Ӧ���ữ�õ���CH3��2C��OH��COOH��M����Է���������86���ȣ�CH3��2C��OH��COOHС18��Ӧ�ǣ�CH3��2C��OH��COOH��Ũ���ᡢ���������·�����������ȥ1����ˮ����F����F����ΪCH2=C��CH3��COOH�� ��̼���������D��
��̼���������D�� ����Ӧ�õ�E��״�����EΪ
����Ӧ�õ�E��״�����EΪ ��˫��A��E��Ӧ�õ�������PC����PCΪ
��˫��A��E��Ӧ�õ�������PC����PCΪ ��
�� ��
����1��a����Ӧ���DZ�ϩ��HBr�����ļӳɷ�Ӧ���ʴ�Ϊ���ӳɷ�Ӧ��
b����Ӧ����±����������ˮ�ⷴӦ����Ӧ�Լ��������ǣ���������ˮ��Һ�����ȣ��ʴ�Ϊ����������ˮ��Һ�����ȣ�
c����Ӧ�۵Ļ�ѧ����ʽ�ǣ�2CH3CH��OH��CH3+O2
| ���� |
| �� |
 +2H2O���ʴ�Ϊ��2CH3CH��OH��CH3+O2
+2H2O���ʴ�Ϊ��2CH3CH��OH��CH3+O2| ���� |
| �� |
 +2H2O��
+2H2O����2��ͨ�����Ϸ���֪��D�DZ��ӣ��ṹ��ʽ��
 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
����3��C��D��Ӧ����˫��A�Ļ�ѧ����ʽ��
 ��
���ʴ�Ϊ��
 ��
����4��F���ж���ͬ���칹�壬��������������F������ͬ���칹�壺�ٺ��м� �ں���̼̼˫�� ���ܷ���������Ӧ ���������࣬Ӧ�Ǽ����γɵ���������������ͬ���칹���У�HCOOCH=CHCH3��
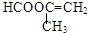 ���ʴ�Ϊ��HCOOCH=CHCH3��
���ʴ�Ϊ��HCOOCH=CHCH3��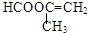 ��
����5��������������֪��E�Ľṹ��ʽ��
 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
���������⿼���л�����ƶ���ϳɣ���ȷ�л���Ĺ����ż��������ǽⱾ��ؼ�����˫��A�ĽṹΪͻ�ƿڲ����������Ʒ����ϵķ����ƶϣ���Ҫѧ���Ը������Ϣ�������ã��Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
���ܸ�ijЩ�������ǽ�����Ӧ�����ܸ�ijЩ����������ǽ���������ֱ�ӷ�Ӧ���ǣ�������
| A��CO |
| B��AgNO3 |
| C��Al2O3 |
| D��NaOH��Һ |
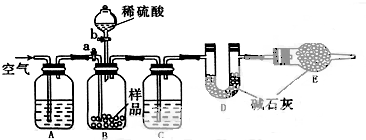
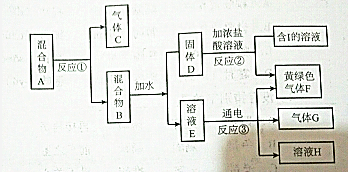

 ��1��2�������飩����Aת��ΪB�Ļ�ѧ����ʽΪ
��1��2�������飩����Aת��ΪB�Ļ�ѧ����ʽΪ