��Ŀ����
�ڽ����к��ȵIJⶨ�У����в�����ȷ����
A. Ϊ��ȷ�ⶨ��Ӧ�����Һ���¶ȣ�ʵ�����¶ȼ�ˮ����Ӧ��С�ձ��ײ��Ӵ�
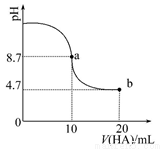
B. ��0.5mol/LNaOH��Һ�ֱ���0.5mol/L�����ᡢ������Һ��Ӧ������ȡ����Һ�����ȣ����õ��к�����ֵ��ͬ
C. ʹ�û��β����������ʹ��Ӧ���Ͼ��ȣ��ӿ췴Ӧ���ʣ���Сʵ�����
D. �ڲⶨ�к���ʵ������Ҫʹ�õ�������������ƽ����Ͳ���ձ������β��������
 ��У����ϵ�д�
��У����ϵ�д����ʵķ�����ж��ַ��������ж��������������ͼ��
��1����ͼ��ʾ�����ʷ����������____________________��
��2����K��Na��H��O��S��N�������ֻ�����Ԫ����ɺ��ʵ����ʣ��ֱ������±��Тڡ��ܡ����档
������� | �� | �� | �� | ������ | �⻯�� |
��ѧʽ | ��HNO3 ��_______ | ��NaOH ��_______ | ��Na2SO4 ��_______ | ��CO2 ��SO3 | ��NH3 |
��3��д�����������Ģ���Һ��Ӧ�����ӷ���ʽ_______________________��
��4��д���������Һ��Ӧ�Ļ�ѧ����ʽ______________________��
��5����ͼ��ijѧУʵ���Ҵӻ�ѧ�Լ��̵���ص�Ũ�����Լ���ǩ�ϵIJ������ݡ����ø�Ũ��������480 mL 1 mol�� L��1��ϡ���ᡣ
�ɹ�ѡ�õ������У��ٽ�ͷ�ιܢ���ƿ���ձ��� ��������ҩ�ע���Ͳ��������ƽ��

��ش��������⣺
a������������ʵ���Ũ��Ϊ __________ mol�� L��1��
b������ϡ����ʱ����ȱ�ٵ������� ______________ (д��������)��
c�������㣬����480mL 1mol�� L��1��ϡ������Ҫ����Ͳ��ȡ����Ũ��������Ϊ_________mL��
d���������Ƶ�ϡ������вⶨ��������Ũ�ȴ���1 mol�� L��1�����ƹ��������и�������������������ԭ���� ___________��
A������ʱ����������ƿ�̶��߽��ж��� ��
B����ϡ�ͺ��ϡ��������ת������ƿ�����žͽ����Ժ��ʵ�������
C��ת����Һʱ��������������Һ��������ƿ���档
D������ƿ������ˮϴ�Ӻ�δ���������������ˮ ��
E�����ݺ�����ƿ����ҡ�Ⱥ���Һ����ڿ̶��ߣ��㲹�伸��ˮ���̶ȴ���


 ��ȡNa��NaOH��H2�ֱ�������ʵ����ĸ�����ǡ�÷�Ӧ����Ӧʱ�ɼ�����У�����Na��NaOH��H2�������ʵ����ʵ���֮��Ϊ
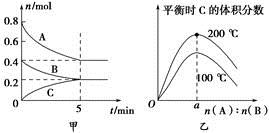
��ȡNa��NaOH��H2�ֱ�������ʵ����ĸ�����ǡ�÷�Ӧ����Ӧʱ�ɼ�����У�����Na��NaOH��H2�������ʵ����ʵ���֮��Ϊ zC(g)��ͼ�ױ�ʾ200 ��ʱ������A��B��C���ʵ�����ʱ��ı仯��ͼ�ұ�ʾ��ͬ�¶���ƽ��ʱC�������������ʼn(A)��n(B)�ı仯��ϵ�������н�����ȷ���ǣ� ����
zC(g)��ͼ�ױ�ʾ200 ��ʱ������A��B��C���ʵ�����ʱ��ı仯��ͼ�ұ�ʾ��ͬ�¶���ƽ��ʱC�������������ʼn(A)��n(B)�ı仯��ϵ�������н�����ȷ���ǣ� ����