��Ŀ����
(10��)��ѧ��ȤС���ijƷ��������Ħ�����ɷּ��京������̽����
�������ϣ�������Ħ������̼��ơ�����������ɣ������������ɷ���������ʱ���������ɡ�
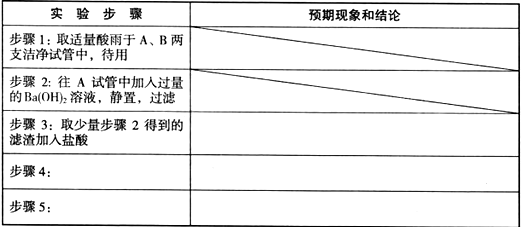
��������Ʒ��̼��ƵĶ����ⶨ��
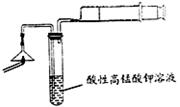
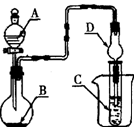
������ͼ��ʾװ�ã�ͼ�мг�������ȥ������ʵ�飬�ⶨC�����ɵ�BaCO3������������ȷ��̼��Ƶ�����������

ʵ�鲽�裺
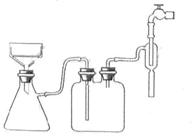
����ͼ��ʾ���Ӻ�װ�ã��ȼ��װ�õ������ԣ�
ȷ��ȡ������Ʒ������4.00g�����ڸ������м�����Ӧ�Լ���
���ɼмף�����ͨ�������Ȼ�����C��
��B�ķ�Һ©���μ����ᷴӦ��ͬʱ�ٴγ�������ͨ�������
��B�в��ٲ��������ֹͣ�μ����
��C�г������ˡ�ϴ�ӡ������BaCO3��
����BaCO3������Ϊ1.97g��
����ʵ����̻ش��������⣺
��1�������ͨ�������������У�______________ __��___ __________��
��2��������м������ϴ�ɾ��ķ����� ��
��3����ʵ��ⶨ�õ�����Ʒ��̼��Ƶ���������Ϊ ������ȡ���д�ʩ���Բⶨ�����ȷ��û��Ӱ�����_____________�����ţ���
a.ʡ�Բ���� b.�μ������һЩ
c.��B��C֮������ʢ�б���NaHCO3��Һ��ϴ��װ��
d. ���ʵ�飬ȡƽ��ֵ����
��4����ͬѧ��Ϊ���زⶨC�����ɵ�BaCO3������ֻҪ�ⶨװ��C������CO2ǰ��������һ������ȷ�ⶨ̼��Ƶ���������������˵���Ƿ���Բ���Ҫ˵���� �� ��
�������ϣ�������Ħ������̼��ơ�����������ɣ������������ɷ���������ʱ���������ɡ�
��������Ʒ��̼��ƵĶ����ⶨ��
������ͼ��ʾװ�ã�ͼ�мг�������ȥ������ʵ�飬�ⶨC�����ɵ�BaCO3������������ȷ��̼��Ƶ�����������

ʵ�鲽�裺
����ͼ��ʾ���Ӻ�װ�ã��ȼ��װ�õ������ԣ�
ȷ��ȡ������Ʒ������4.00g�����ڸ������м�����Ӧ�Լ���
���ɼмף�����ͨ�������Ȼ�����C��
��B�ķ�Һ©���μ����ᷴӦ��ͬʱ�ٴγ�������ͨ�������
��B�в��ٲ��������ֹͣ�μ����
��C�г������ˡ�ϴ�ӡ������BaCO3��
����BaCO3������Ϊ1.97g��
����ʵ����̻ش��������⣺
��1�������ͨ�������������У�______________ __��___ __________��
��2��������м������ϴ�ɾ��ķ����� ��
��3����ʵ��ⶨ�õ�����Ʒ��̼��Ƶ���������Ϊ ������ȡ���д�ʩ���Բⶨ�����ȷ��û��Ӱ�����_____________�����ţ���
a.ʡ�Բ���� b.�μ������һЩ
c.��B��C֮������ʢ�б���NaHCO3��Һ��ϴ��װ��
d. ���ʵ�飬ȡƽ��ֵ����
��4����ͬѧ��Ϊ���زⶨC�����ɵ�BaCO3������ֻҪ�ⶨװ��C������CO2ǰ��������һ������ȷ�ⶨ̼��Ƶ���������������˵���Ƿ���Բ���Ҫ˵���� �� ��
��1������B��C�еķ�Ӧ�1�֣� �����ɵ�CO2����ȫ������C�У�ʹ֮��ȫ��Ba(OH)2��Һ���գ�1�֣�
��2��ȡ�����Һ������ϡ���ᣨ������������Һ�������ް�ɫ�������֣������ϴ�ɾ���������AgNO3��Һ����Cl��Ҳ���֣���2�֣�
��3��25%��2�֣� d��2�֣�
��4�������� ������õĽ����ƫ����ΪB�е�ˮ������HCl��������װ��C��ʹ��CO2�������ƫ��2�֣�
��2��ȡ�����Һ������ϡ���ᣨ������������Һ�������ް�ɫ�������֣������ϴ�ɾ���������AgNO3��Һ����Cl��Ҳ���֣���2�֣�
��3��25%��2�֣� d��2�֣�
��4�������� ������õĽ����ƫ����ΪB�е�ˮ������HCl��������װ��C��ʹ��CO2�������ƫ��2�֣�
��1�������ͨ��������һ�������ʹ��Ӧ������ֻ�Ͼ��ȣ�ʹ��Ӧ��֮���ַ�Ӧ����������ã�ͬʱ����ͨ������������ʹ��Ӧ���ɵ�CO2�����ֱ�Cװ���е�Ba(OH)2���գ������ʵ���ȷ�ȡ�
��2������Cװ���е�Ba(OH)2��Һ�ǹ���������Һ�к���CL����������BaCO3������渽��Ba2+��OH����CL�������ԣ�������м������ϴ�ɾ��ķ�����Ϊ����Ba2+��OH����CL��������Ba2+���ӵķ����ɼ��뺬�����Ե������λ�ϡ���ᣬ���а�ɫ������δϴ�Ӹɾ�����û�а�ɫ������ϴ�Ӹɾ�����Ȼ��Ҳ����ͨ������CL���ķ�ʽ�����������ϡ���ᣬȻ�����������Һ�����а�ɫ���������������δϴ�Ӹɾ�����û�а�ɫ���������������ϴ�Ӹɾ���
��3��1.97gBaCO3�����ʵ���Ϊ0.01 mol������CԪ���غ㣬�ɵ�CaCO3������Ϊ1g�����Կɵ���Ʒ��CaCO3����������Ϊ25%��
���ʡ���˲��貽��ۣ�����ʹʵ��ⶨ���ƫ�ߣ���Ϊ��Ӧǰװ���еĿ����к��ж�����̼���壬���Ի�ʹʵ��ⶨ���ƫ�ߡ�����μ������һЩ����ʹ��Ӧ���ʹ��죬�������ɵĶ�����̼���岻�ܳ�ֱ�����������Һ���գ������ʹʵ��ⶨ���ƫ�͡������B��C֮������ʢ�б���NaHCO3��Һ��ϴ��װ�ã���ʹʵ��ⶨ���ƫ�ߣ���Ϊ���ɵĶ�����̼�����л�����������HCL���壬HCL�������NaHCO3��Һ��Ӧ�����ɶ�����̼���壬ʹ������̼�������࣬�������ʵ��ⶨ���ƫ�ߡ������ʵ�飬ȡƽ��ֵ����ֻ�����ʵ���ȷ�ȡ�
��4�������� ������õĽ����ƫ����ΪB�е�ˮ������HCl��������װ��C��ʹ��CO2�������ƫ����ʵ��ⶨ���ƫ��
��2������Cװ���е�Ba(OH)2��Һ�ǹ���������Һ�к���CL����������BaCO3������渽��Ba2+��OH����CL�������ԣ�������м������ϴ�ɾ��ķ�����Ϊ����Ba2+��OH����CL��������Ba2+���ӵķ����ɼ��뺬�����Ե������λ�ϡ���ᣬ���а�ɫ������δϴ�Ӹɾ�����û�а�ɫ������ϴ�Ӹɾ�����Ȼ��Ҳ����ͨ������CL���ķ�ʽ�����������ϡ���ᣬȻ�����������Һ�����а�ɫ���������������δϴ�Ӹɾ�����û�а�ɫ���������������ϴ�Ӹɾ���
��3��1.97gBaCO3�����ʵ���Ϊ0.01 mol������CԪ���غ㣬�ɵ�CaCO3������Ϊ1g�����Կɵ���Ʒ��CaCO3����������Ϊ25%��
���ʡ���˲��貽��ۣ�����ʹʵ��ⶨ���ƫ�ߣ���Ϊ��Ӧǰװ���еĿ����к��ж�����̼���壬���Ի�ʹʵ��ⶨ���ƫ�ߡ�����μ������һЩ����ʹ��Ӧ���ʹ��죬�������ɵĶ�����̼���岻�ܳ�ֱ�����������Һ���գ������ʹʵ��ⶨ���ƫ�͡������B��C֮������ʢ�б���NaHCO3��Һ��ϴ��װ�ã���ʹʵ��ⶨ���ƫ�ߣ���Ϊ���ɵĶ�����̼�����л�����������HCL���壬HCL�������NaHCO3��Һ��Ӧ�����ɶ�����̼���壬ʹ������̼�������࣬�������ʵ��ⶨ���ƫ�ߡ������ʵ�飬ȡƽ��ֵ����ֻ�����ʵ���ȷ�ȡ�
��4�������� ������õĽ����ƫ����ΪB�е�ˮ������HCl��������װ��C��ʹ��CO2�������ƫ����ʵ��ⶨ���ƫ��

��ϰ��ϵ�д�
�����Ŀ
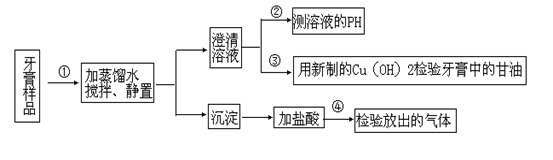
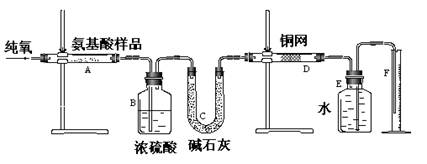
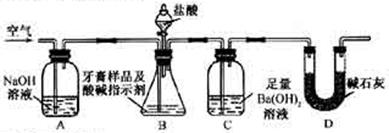
 ��죬�ּ�����ʹ�����ǵ�ľ����ȼ������0.28L������£���
��죬�ּ�����ʹ�����ǵ�ľ����ȼ������0.28L������£��� ��ijƷ��������Ħ�����ɷּ��京����������̽����
��ijƷ��������Ħ�����ɷּ��京����������̽���� __________________________________________��
__________________________________________��
 ___��
___��