��Ŀ����
13����ͼ��ʾ�绯ѧװ���е缫��Ϊ���Ե缫������������SO2�������ų�����Һ����������NO2������������Ⱦ������˵����ȷ���ǣ�������
| A�� | ���е����ӽ���Ĥ��ѡ�������ӽ���Ĥ | |
| B�� | �����ĵ缫��ӦʽΪ��2HSO3-+2e-=S2O42-+2OH- | |
| C�� | �����ĵ缫��ӦʽΪ��SO2+2H2O-2e-=SO42-+4H+ | |
| D�� | ��Ӧһ��ʱ����Ե缫�������缫���Ե缫���ﲻ����Ӱ�� |
���� ����ͼʾ��֪��������������Ϊ����������Զ����������ڵ���Ϊ��������������������ӦSO2-2e-+2H2O�TSO42-+4H+���������Դ������a������bΪ��Դ�����������ĵ缫��ӦʽΪ��2HSO3-+2H++2e-�TS2O42-+2H2O��������ӵ��ƶ��������ķ���������
��� �⣺A������������ӦSO2-2e-+2H2O�TSO42-+4H+��������Ӧ��Ҫ����H+�������е����ӽ���ĤӦѡ�������ӽ���Ĥ����A����
B��bΪ��Դ���������Դ���������ĵ缫Ϊ������������ӦΪHSO3-�õ���������S2O42-���������ĵ缫��ӦʽΪ��2HSO3-+2H++2e-�TS2O42-+2H2O����B����
C���������Դ������a������������ӦΪSO2�õ����ӣ�����SO42-���������ĵ缫��ӦʽΪ��SO2+2H2O-2e-=SO42-+4H+����C��ȷ��
D��Fe�缫�������Ե缫����û��Ӱ�죬����Ϊ��������Ϊ���Ե缫���ᷢ���缫��Ӧ��Ӱ��缫�����D����
��ѡC��
���� ���⿼���˵��ԭ���ķ���Ӧ�ã���Ҫ�ǵ缫��Ӧ���缫�����жϣ��������ԭ���ǹؼ�����Ŀ�Ѷ��еȣ����е��⣮

��ϰ��ϵ�д�
�����Ŀ
4������˵����ȷ���ǣ�������
| A�� | �¶ȸߵ��������ܲ�һ��������ƽ������һ���� | |
| B�� | ������������Ϊ����ʱ��������������Ӽ����ļ�С������ | |
| C�� | ����������������������һ������ | |
| D�� | �����Ӽ�ľ�������ʱ��������һ����С |
1��������������Ӱ�����ǵ�������е���������������������������������Ҫԭ��֮һ������������������������ж��ַ�����
��1���û���̿��ԭ�����Դ����������ij�о�С����ij�ܱ������м���һ�����Ļ���̿��NO��������Ӧ��C��s��+2NO��g��?N2��g��+CO2��g����H=Q kJ•mol-1����T1��ʱ����Ӧ���е���ͬʱ����
�����ʵ�Ũ�����£�
��0��10min�ڣ�NO��ƽ����Ӧ����v��NO��=0.042mol/��L•min����T1��ʱ���÷�Ӧ��ƽ�ⳣ��K=$\frac{9}{16}$��
��30min��ֻ�ı�ijһ��������Ӧ���´ﵽƽ�⣬���ݱ��е������жϸı������������bc ����
��ĸ����
a������һ�����Ļ���̿ b��ͨ��һ������NO
c���ʵ���С��������� d��������ʵĴ���
����30min�������¶���T2�棬�ﵽƽ��ʱ�������У�NO��N2��CO2��Ũ��֮��Ϊ5��3��3����Q�������������=��������0��
��2��NH3����ԭ�������SCR��������ĿǰӦ����㷺���������������ѳ���������Ӧԭ����ͼ��ʾ��

����ͼ��֪��SCR�����е�������ΪNO��NO2����֪c��NO2����c��NO��=1��1ʱ�ѵ�Ч����ѣ�
������1mol N2ʱ��Ӧ�ų�������ΪQ kJ����ʱ��Ӧ���ѵ���Ӧ���Ȼ�ѧ����ʽΪ2NH3��g��+NO��g��+NO2��g��=2N2��g��+3H2O��g����H=-2QkJ•mol-1
��ͼ���Dz�ͬ����Mn��Cr�ڲ�ͬ�¶��¶�Ӧ���ѵ��ʣ���ͼ��֪��ҵʹ�õ���ѵĴ�������
Ӧ���¶ȷֱ�ΪMn��200�����ң�
��3������β���е�SO2����ʯ��ˮ�����գ��������������Һ����֪������Ka1��H2SO3��=1.8��10-2��Ka2��H2SO3��=6.0��10-9�������£����ij��CaSO3��ˮ�γɵ���ҺpH=9������SO32-�ĵڶ���ˮ�⣬��Ksp��CaSO3��=4.2��10-9��
��1���û���̿��ԭ�����Դ����������ij�о�С����ij�ܱ������м���һ�����Ļ���̿��NO��������Ӧ��C��s��+2NO��g��?N2��g��+CO2��g����H=Q kJ•mol-1����T1��ʱ����Ӧ���е���ͬʱ����
�����ʵ�Ũ�����£�
| ʱ�䣨min�� Ũ�ȣ�mol•L-1�� | 0 | 10 | 20 | 30 | 40 | 50 |
| NO | 1.00 | 0.58 | 0.40 | 0.40 | 0.48 | 0.48 |
| N2 | 0 | 0.21 | 0.30 | 0.30 | 0.36 | 0.36 |
| CO2 | 0 | 0.21 | 0.30 | 0.30 | 0.36 | 0.36 |
��30min��ֻ�ı�ijһ��������Ӧ���´ﵽƽ�⣬���ݱ��е������жϸı������������bc ����
��ĸ����
a������һ�����Ļ���̿ b��ͨ��һ������NO
c���ʵ���С��������� d��������ʵĴ���
����30min�������¶���T2�棬�ﵽƽ��ʱ�������У�NO��N2��CO2��Ũ��֮��Ϊ5��3��3����Q�������������=��������0��
��2��NH3����ԭ�������SCR��������ĿǰӦ����㷺���������������ѳ���������Ӧԭ����ͼ��ʾ��

����ͼ��֪��SCR�����е�������ΪNO��NO2����֪c��NO2����c��NO��=1��1ʱ�ѵ�Ч����ѣ�
������1mol N2ʱ��Ӧ�ų�������ΪQ kJ����ʱ��Ӧ���ѵ���Ӧ���Ȼ�ѧ����ʽΪ2NH3��g��+NO��g��+NO2��g��=2N2��g��+3H2O��g����H=-2QkJ•mol-1
��ͼ���Dz�ͬ����Mn��Cr�ڲ�ͬ�¶��¶�Ӧ���ѵ��ʣ���ͼ��֪��ҵʹ�õ���ѵĴ�������
Ӧ���¶ȷֱ�ΪMn��200�����ң�
��3������β���е�SO2����ʯ��ˮ�����գ��������������Һ����֪������Ka1��H2SO3��=1.8��10-2��Ka2��H2SO3��=6.0��10-9�������£����ij��CaSO3��ˮ�γɵ���ҺpH=9������SO32-�ĵڶ���ˮ�⣬��Ksp��CaSO3��=4.2��10-9��
8����1��һ�������£�CO2��1.0mol•L-1NaOH��Һ��ַ�Ӧ�ų����������һ��
��һ��
��������CO2��NaOH��Һ��Ӧ����NaHCO3���Ȼ�ѧ����ʽΪNaOH��aq��+CO2��g���TNaHCO3��aq����H=-��4x-y��kJ/mol ��
��2���ý�̿��ԭNO�ķ�ӦΪ��2NO��g��+C��s��?N2��g��+CO2��g�������ݻ���Ϊ1L�ļס��ҡ����������������зֱ���������Ľ�̿��һ������NO����ø�������n��NO���淴Ӧʱ��t�ı仯����������
������
�ټ������У�0��40min����NO��Ũ�ȱ仯��ʾ��ƽ����Ӧ����v��NO��=0.0125mol/��L•min����
�ڸ÷�Ӧ�ġ�Hb������ţ�
a������0 b��С��0 c������0d������ȷ��
�۱������ﵽƽ��ʱ��NO��ת����Ϊ60% ��
��3��298Kʱ��NH3•H2O�ĵ��볣��Kb=2��10-5��H2CO3�ĵ��볣��Kal=4��10-7��Ka2=4��10-11����NH4HCO3��Һ�У�c��NH4+����c��HCO3-�������������������=��������ӦNH4++HCO3-+H2O?NH3•H2O+H2CO3��ƽ�ⳣ��K����ֵ���ÿ�ѧ��������ʾ��Ϊ1.25��10-3��
��һ��
| ��Ӧ��� | CO2�����ʵ���/mol | NaOH��Һ�����/L | �ų�������/KJ |
| 1 | 0.5 | 0.75 | x |
| 2 | 1.0 | 2.00 | y |
��2���ý�̿��ԭNO�ķ�ӦΪ��2NO��g��+C��s��?N2��g��+CO2��g�������ݻ���Ϊ1L�ļס��ҡ����������������зֱ���������Ľ�̿��һ������NO����ø�������n��NO���淴Ӧʱ��t�ı仯����������
������
| �¶� t/min | 0 | 40 | 80 | 120 | 160 |
| �ף�673K�� | 2.00 | 1.50 | 1.10 | 0.80 | 0.80 |
| �ң�T�� | 2.00 | 1.45 | 1.00 | 1.00 | 1.00 |
| ����673K�� | 1.00 | 0.80 | 0.65 | 0.53 | 0.45 |
�ڸ÷�Ӧ�ġ�Hb������ţ�
a������0 b��С��0 c������0d������ȷ��
�۱������ﵽƽ��ʱ��NO��ת����Ϊ60% ��
��3��298Kʱ��NH3•H2O�ĵ��볣��Kb=2��10-5��H2CO3�ĵ��볣��Kal=4��10-7��Ka2=4��10-11����NH4HCO3��Һ�У�c��NH4+����c��HCO3-�������������������=��������ӦNH4++HCO3-+H2O?NH3•H2O+H2CO3��ƽ�ⳣ��K����ֵ���ÿ�ѧ��������ʾ��Ϊ1.25��10-3��
18��${\;}_{53}^{131}$I�dz�����ѱ����֮һ������ͨ���ⶨ������ˮ��${\;}_{53}^{131}$I�ĺ����仯�����˵�վ�Ƿ���й¶�������й�${\;}_{53}^{131}$I�������в���ȷ���ǣ�������
| A�� | ${\;}_{53}^{131}$I�Ļ�ѧ������${\;}_{53}^{127}$��ͬ | |
| B�� | ${\;}_{53}^{131}$I��ԭ������Ϊ53 | |
| C�� | ${\;}_{53}^{131}$I�ĺ��������Ϊ78 | |
| D�� | ${\;}_{53}^{131}$Iԭ�Ӻ������������������� |
5�����������У��Ⱥ����Ӽ��ֺ����ۼ����ǣ�������
| A�� | H2O | B�� | NaOH | C�� | H2 | D�� | Na2O |
2�����������У���Ԫ�ػ��ϼ���͵��ǣ�������
| A�� | Cl2 | B�� | HCl | C�� | HClO | D�� | KClO3 |
3�����ǻ������л�����ɵĻ������У����ڴ�����ǣ�������
| A�� | NH2- | B�� |  | C�� | 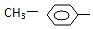 | D�� | R-CO- |