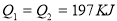��Ŀ����
��16�֣����ݻ�Ϊ1.00 L�������У�ͨ��һ������N2O4��������ӦN2O4(g)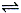 2NO2(g)�����¶����ߣ�����������ɫ����ش��������⣺
2NO2(g)�����¶����ߣ�����������ɫ����ش��������⣺
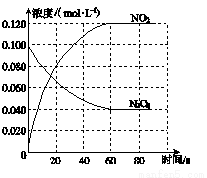
��1����Ӧ�Ħ�H________0(����ڡ���С�ڡ�)��100 ��ʱ����ϵ�и��� ��Ũ����ʱ��仯��ͼ��ʾ����0��60 sʱ�Σ���Ӧ����v(N2O4)Ϊ________mol��L��1��s��1����Ӧ��ƽ�ⳣ��K1Ϊ________��
��2��100 ��ʱ��ƽ��ı䷴Ӧ�¶�ΪT��c(N2O4)��0.002 0 mol��L��1��s��1��ƽ�����ʽ��ͣ���10 s�ִﵽƽ�⡣
��T________100 ��(����ڡ���С�ڡ�)���ж�������____________________________��
����ʽ�����¶�Tʱ��Ӧ��ƽ�ⳣ��K2��_________________________________________��
��3���¶�Tʱ��Ӧ��ƽ�����Ӧ�������ݻ�����һ�룬ƽ����________(�����Ӧ�����淴Ӧ��)�����ƶ����ж�������_________________________________________________________��
��16�֣���1������ 0.001 0 0.36 mol��L��1
��2���ٴ��� ��Ӧ���������ȣ���Ӧ�����ȷ�����У����¶�����
��ƽ��ʱ��c(NO2)��0.120 mol��L��1��0.002 0 mol��L��1��s��1��10 s��2��0.16 mol��L��1
c(N2O4)��0.040 mol��L��1��0.002 0 mol��L��1��s��1��10 s��0.020 mol��L��1
K2��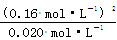 ��1.3 mol��L��1
��1.3 mol��L��1
��3���淴Ӧ ���������������ķ�Ӧ������ѹǿƽ�����淴Ӧ�����ƶ�
��������
�����������1���¶����ߣ�����������ɫ���˵�������¶ȣ�ƽ�������ɶ��������ķ����ƶ�������ƽ�������ƶ��������������ȷ�Ӧ����H>0����0��60 sʱ�Σ�N2O4�����ʵ���Ũ�ȴ�0.100mol/L���ٵ�0.040mol/L�����Է�Ӧ����v(N2O4)=��0.100-0.040��mol/L/60s=0.001 mol��L��1��s��1��ƽ��ʱc��N2O4��=0.040mol/L��c��NO2��=0.120mol/L������100���ƽ�ⳣ��K= c��NO2��2/ c��N2O4��=0.36 mol��L��1
��2����c(N2O4)��0.002 0 mol��L��1��s��1��ƽ�����ʽ��ͣ�˵��ƽ�������ƶ������¶����ߣ�T>100�棻��Ϊ����Ӧ�����ȷ�Ӧ���¶����ߣ�ƽ����������ƶ���
��c(N2O4)��0.002 0 mol��L��1��s��1��ƽ�����ʽ��ͣ���10 s�ִﵽƽ�⣬��10s��ﵽƽ��ʱ��c(N2O4)=0.040mol/L-0.002 0 mol��L��1��s��1��10s=0.02mol/L�����ʱc��NO2��=0.120mol/L+2��0.02mol/L=0.16mol/L������T�¶ȵ�ƽ�ⳣ��K=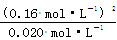 ��1.3 mol��L��1
��1.3 mol��L��1
��3���¶�Tʱ��Ӧ��ƽ�����Ӧ�������ݻ�����һ�룬��ѹǿ����ƽ�������ƶ�����Ϊ����ѹǿ��ƽ�����������ʵ������ٵķ����ƶ�������������������ʵ������ٵķ�������ƽ�������ƶ���
���㣺����ƽ���ƶ����жϣ���ѧƽ�ⳣ������Ӧ���ʵļ���

 2HI(g) ����֪H2��I2����ʼŨ�Ⱦ�Ϊ0.1 mol��L��1ʱ����ƽ��ʱHIŨ��Ϊ0.16 mol��L��1����H2��I2����ʼŨ�Ⱦ���Ϊ0.2 mol��L��1����ƽ��ʱH2��Ũ�ȣ�mol��L��1����
2HI(g) ����֪H2��I2����ʼŨ�Ⱦ�Ϊ0.1 mol��L��1ʱ����ƽ��ʱHIŨ��Ϊ0.16 mol��L��1����H2��I2����ʼŨ�Ⱦ���Ϊ0.2 mol��L��1����ƽ��ʱH2��Ũ�ȣ�mol��L��1���� 2SO3(g) ��H��-197 kJ��mol��1 ������ͬ���¶��£����ܱ����������ͨ��2molSO2��1molO2���ﵽƽ��ʱ�ų�����Q1�� ����һ��ͬ������ܱ����������ͨ��1molSO2��0.5molO2���ﵽƽ��ʱ�ų�����Q2�������й�ϵ����ȷ����
2SO3(g) ��H��-197 kJ��mol��1 ������ͬ���¶��£����ܱ����������ͨ��2molSO2��1molO2���ﵽƽ��ʱ�ų�����Q1�� ����һ��ͬ������ܱ����������ͨ��1molSO2��0.5molO2���ﵽƽ��ʱ�ų�����Q2�������й�ϵ����ȷ����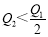 B.
B.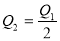 C.
C.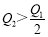 D.
D.