��Ŀ����
18��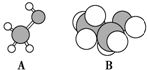 ��ͼ��A���ӵ����ģ�ͺ�B���ӵı���ģ�ͣ��ش��������⣺
��ͼ��A���ӵ����ģ�ͺ�B���ӵı���ģ�ͣ��ش��������⣺��1��A��B�Ĺ�ϵ��ͬϵ�
��2��д��A�����ڴ������������¼��Ⱥ�������Ӧ�Ļ�ѧ����ʽ2CH3OH+O2$��_{��}^{����}$2HCHO+2H2O��
��3��д��B���Ӻͽ����Ʒ�Ӧ�Ļ�ѧ����ʽ2Na+2CH3CH2OH-��2CH3CH2ONa+H2����
��4��B�ڼ����������ܹ���HBr������Ӧ���������飬�÷�Ӧ������ȡ����Ӧ��
���� �ɽṹģ�Ϳ�֪AΪ�״���BΪ�Ҵ����������ǻ����ɷ���ȡ����������Ӧ�����Ҵ���������ȥ��Ӧ���Դ˽����⣮
��� �⣺�ɽṹģ�Ϳ�֪AΪ�״���BΪ�Ҵ���
��1��AΪCH3OH��BΪCH3CH2OH���ṹ���ƣ�������������1��CH2ԭ���ţ���������ͬϵ��ʴ�Ϊ��ͬϵ�
��2���Ҵ�����������ȩ����Ӧ����ʽΪ��2CH3OH+O2$��_{��}^{����}$2HCHO+2H2O���ʴ�Ϊ��2CH3OH+O2$��_{��}^{����}$2HCHO+2H2O��
��3���Ҵ����Ʒ�Ӧ�����Ҵ��ƺ�����������ʽΪ2Na+2CH3CH2OH��2CH3CH2ONa+H2�����ʴ�Ϊ��2Na+2CH3CH2OH��2CH3CH2ONa+H2����
��4���Ҵ��ڼ����������ܹ���HBr������Ӧ���������飬��Ӧ�ķ���ʽΪCH3CH2OH+HBr$\stackrel{��}{��}$CH3CH2Br+H2O���ʴ�Ϊ��CH3CH2OH+HBr$\stackrel{��}{��}$CH3CH2Br+H2O���ʴ�Ϊ��ȡ����Ӧ��
���� ���⿼���л���Ľṹ�����ʣ�Ϊ�߿���Ƶ���㣬���սṹ�й����������ʵĹ�ϵΪ���Ĺؼ������ش����ƶϼ����ʵĿ��飬��Ŀ�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
2��XԪ�����γ�H2X��XO2���ֻ������Ԫ�ص�ԭ�������ǣ�������
| A�� | 13 | B�� | 14 | C�� | 15 | D�� | 16 |
9����NA��ʾ�����ӵ�������ֵ������˵����ȷ���ǣ�������
| A�� | ���³�ѹ�£�4.4gCO2��N2O��������к��е�ԭ������Ϊ0.3NA | |
| B�� | ��״���£�11.2 L H2O�к��е���ԭ����ΪNA | |
| C�� | H2O2�ֽ����1molO2��ת�Ƶĵ�����Ϊ4NA | |
| D�� | 1 mol•L-1 CuCl2��Һ�к��е���������Ϊ2NA |
3�����б仯�����������������ʵ�ֵ��ǣ�������
| A�� | CuO��CuSO4 | B�� | CO2��CO | C�� | HNO3��NO2 | D�� | Fe��Fe3O4 |
10��ʵ���ҿ�������ˮ�����ƹ���ͼ�ʯ�Ҽ�ǿ����ȡ�������壮������һ����ɫ����ζ��������ˮ�����壮�ɲ��õķ���װ�ú��ռ������ֱ��ǣ�������
���������ķ���װ�� ���ƶ�����̼�ķ���װ�� ����ˮ���������������������� ������������������
���������ķ���װ�� ���ƶ�����̼�ķ���װ�� ����ˮ���������������������� ������������������
| A�� | �ڡ��ۻ�� | B�� | �ڡ��ۻ�� | C�� | �١��ۻ�� | D�� | �١��ۻ�� |
7�����й����С�H��������ǣ�������
| A�� | ��������ȷֽ������� | B�� | ���ȷ�Ӧ | ||
| C�� | �ƺ�ˮ��Ӧ | D�� | ʵ�����Ʊ����� |