��Ŀ����
9�� ��������������ĸ�ҹ���������������ƣ�NaNO2���������س��������ʳ�õ�˵����ij��ȤС�鿪չ����̽�����
��������������ĸ�ҹ���������������ƣ�NaNO2���������س��������ʳ�õ�˵����ij��ȤС�鿪չ����̽������һ����������
NaNO2�ж������н�ǿ�����Ժͽ�����ԭ�ԣ�NaNO2��Һ�Լ��ԣ����ҹ涨����Ʒ��NaNO2�������ܳ���30mg/kg��
��1��NaNO2��Һ�Լ��Ե�ԭ����NO2-+H2O?HNO2+OH-���������ӷ���ʽ���
���������NaNO2��ʳ��
��2������ϡ���ᡢ���ۺ�KI��Һ������Һ��������NaNO2�����ѧʽ��
�����̽��NaNO3��NaNO2���ת��
��3����ȡNaNO2��NaNO3�����Һ��װ��ʾ��ͼ��ͼ��
��֪��2NO2+2NaOH�TNaNO2+NaNO3+H2 O
��װ��II�е��Լ�����Ϊˮ�������Ƕ���������ˮ��Ӧ�ܲ���һ���������ŵ������л������Ⱦ��
��װ��III�з�Ӧ��ȫ�������IJ����Ǵ���a��ѡ�a����b������ͬ�����رջ���b��
��4����NaNO2��NaNO3�����Һ¶���ڿ�������ҹ�����Һ��pH��С������Һ��NaNO2�ĺ������٣�ѡ����ࡱ�������١������䡱����ԭ�������2NaNO2+O2=2NaNO3���û�ѧ����ʽ���ͣ���
��ģ����������NaNO2�����ı仯
��5���ֱ�� 1000g���������� 1000g��ҹ��������ȡNaNO2�������Һ���ٷֱ���0.00500mol/L���Ը��������Һ�ζ����������������12.00mL����ҹ��������16.00mL������ʾ��MnO4һת��ΪMn2+��NO2һת��ΪNO3-��
���ڸõζ�ʵ������Ҫ�õ���ʵ��������BE
A������ƿ B����ʽ�ζ��� C����ʽ�ζ��� D����Һ©�� E����ƿ F�� ��ƿ
�ڵζ��յ���ж�����Ϊ���������һ�α�Һʱ����ƿ����Һ��죬��30���ڲ���ɫ��
�۸�ҹ������NaNO2�ĺ�����13.8mg/kg��������˵������õ��ĸ�ҹ�����ܣ�ѡ��ܡ����ܡ���ʳ�ã�
���� ��1��NaNO2Ϊǿ�������Σ�������������ܷ���ˮ�⣻
��2��NaNO2�������ԣ��ܽ������������ɵⵥ�ʣ��ݴ˴��⣻
��3����װ��IIΪβ������װ�ã�����������ˮ��Ӧ����һ����������������Ⱦ������
��װ��III�з�Ӧ��ȫ��Ҫ��װ���еĶ���������װ��II���գ�����Ҫ�ȴ�a���ٹر�b��
��4����NaNO2��Һ¶���ڿ����У��ܱ������е����������������ƣ��������ˮ����������������Ӽ��٣�������Һ��pH��С���ݴ˴��⣻
��5���ٸ���������ԭ�ζ��IJ�����ȷ������������
����Ϊ������ص�ϡ��Һ�Ǻ�ɫ�ģ����Եζ��յ�ʱ����Һ����ֺ�ɫ��
�۸��ݵ��ӵ�ʧ�غ��֪��ϵʽ2KMnO4��5NaNO2���ɹ�ϵʽ�������ĵĸ��������Һ�����ʵ����ɼ�����������Ƶ�����������ȷ����ҹ������NaNO2�ĺ��������ݴ��ж��ܷ�ʳ�ã�
��� �⣺��1��NaNO2Ϊǿ�������Σ�������������ܷ���ˮ�⣬ˮ������ӷ���ʽΪNO2-+H2O?HNO2+OH-��
�ʴ�Ϊ��NO2-+H2O?HNO2+OH-��
��2��NaNO2�������ԣ��ܽ������������ɵⵥ�ʣ�����Ҫ���������£�NaNO2��ʹ���ۡ�KI��Һ������
�ʴ�Ϊ��NaNO2��
��3���٢�װ���в����Ķ�����������ˮ��Ӧ�ܲ���һ���������ŵ������л������Ⱦ������ˮ������Ϊװ�â��е��Լ���
�ʴ�Ϊ������������ˮ��Ӧ�ܲ���һ���������ŵ������л������Ⱦ��
��װ��III�з�Ӧ��ȫ��Ҫ��װ���еĶ���������װ��II���գ�����Ҫ�ȴ�a���ٹر�b��
�ʴ�Ϊ��a��b��
��4����NaNO2��Һ¶���ڿ����У��ܱ������е����������������ƣ���Һ��NaNO2�ĺ������������ˮ����������������Ӽ��٣�������Һ��pH��С����Ӧ�Ļ�ѧ����ʽΪ2NaNO2+O2=2NaNO3��
�ʴ�Ϊ�����٣�2NaNO2+O2=2NaNO3��
��5���ٸ���������ԭ�ζ��IJ�����֪���õ�������Ϊ��ʽ�ζ��ܡ���ƿ����ѡBE��
����Ϊ������ص�ϡ��Һ�Ǻ�ɫ�ģ����Եζ��յ�ʱ����Һ����ֺ�ɫ�����Եζ��յ���ж�����Ϊ�����������һ�α�Һʱ����ƿ����Һ��죬��30���ڲ���ɫ��
�ʴ�Ϊ�����������һ�α�Һʱ����ƿ����Һ��죬��30���ڲ���ɫ��
�۸��������֪�����ҹ��������ȡ��NaNO2��Ӧ��KMnO4�����ʵ���Ϊ0.00500mol/L��0.016L=8.0��10-5 mol�����ݵ��ӵ�ʧ�غ��֪��ϵʽ2KMnO4��5NaNO2���ɹ�ϵʽ��֪�������Ƶ�����Ϊ$\frac{5}{2}$��8.0��10-5 mol��69g/mol=13.8mg�����Ը�ҹ������NaNO2�ĺ���Ϊ$\frac{13.8mg}{1Kg}$=13.8mg/Kg��30mg/kg��������ʳ�ã�
�ʴ�Ϊ��13.8���ܣ�
���� ������Ҫ����ʵ�����������ʵ��ԭ����Ӧ�÷������е��Ѷȣ�����ʱע���Ԫ�ػ�����֪ʶ��ʵ��ԭ����ֽ�ϣ�ע�⻯ѧ�������ȷ��д��

| A�� | ��������Դ����̫���ܡ����ܵȣ����ٶԻ�ʯ��Դ��������Ҳ���ٶԴ�������Ⱦ | |
| B�� | ֱ���ӽ���2.5��10-6m�Ŀ����PM2.5����ɢ�ڿ������γɵĻ������ж����ЧӦ | |
| C�� | ʵʩ��ú����������ú�ĵ硱�����ȼ�ϸ��칤�̣������ڱ������� | |
| D�� | ���ͷ���������ʱ�����һ������NO��CO��������Ԫ��װ�ÿ��Լ�����Ⱦ����ŷ� |
| A�� | �Է���Ӧһ�����������Է���Ӧһ�����ؼ�С�� | |
| B�� | Na2CO3��Һ��ˮϡ�ͺָ���ԭ�¶ȣ�pH��Kw����С | |
| C�� | pH=5��CH3COOH��Һ��PH=5��NH4Cl��Һ�У�c��H+������� | |
| D�� | ��Na2S��Һ�м���AgCl���壬��Һ��c��S2-���½� |
| A | B | C | D | |
| ǿ����� | Fe | NaCl | CaCO3 | HNO3 |
| ������� | CH3COOH | NH3 | H2CO3 | Fe��OH��3 |
| �ǵ���� | ���� | BaSO4 | C2H5OH | H2O |
| A�� | A | B�� | B | C�� | C | D�� | D |
| ѡ�� | ʵ�鷽�� | ���� |
| A | ��ȥ�屽�е������嵥�ʣ���NaOH��Һϴ�ӡ���Һ | ����Br2������NaOH��Һ��Ӧ |
| B | �����Ҵ��������������������ƣ����������Ҵ����ټ������Ũ���ᣬ������������ | ����ֱ�����ɷ���õ��������Ҵ������� |
| C | ��ȡ���͵�ˮ�еĵⵥ�ʣ����Ҵ���ȡ | ��ȷ�����ʵ��������Ҵ����л��ܼ� |
| D | ��ȥ�����л��е�������ϩ������ˮϴ�� | ��ȷ����ϩ������ˮ�����ӳɷ�Ӧ |
| A�� | A | B�� | B | C�� | C | D�� | D |
��2��Zn��s��+Ag2O��s���TZnO��s��+2Ag��s������H=-317.3kJ/mol
��2Ag��s��+$\frac{1}{2}$O2��g���TAg2O��s���ġ�H���ڣ�������
| A�� | 31.0kJ/mol | B�� | -665.6kJ/mol | C�� | 332.8 kJ/mol | D�� | -31.0 kJ/mol |
| A�� | ˮ�ࡢ������ˮ�����ﶼ�ǹ�������Ʒ | |
| B�� | �����£������Թ�����ɽ�̿��ʯӢɰ��SiO2����ȡ��ķ�Ӧ | |
| C�� | ���ά����Ҫ�ɷ���SiO2 | |
| D�� | ��Ȼ���й�Ԫ�ص������ḻ�������ڴ����ĵ��ʹ� |
| A�� | ����ͱ��� | B�� |  �� ��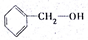 | C�� | 2-������1-���� | D�� | 2-��ϩ�ͱ�ϩ |
| A�� | ��ʯ--SiO2 | B�� | ������--FeS2 | ||
| C�� | ����--NaOH | D�� | â��--Na2SO4•10H2O |