��Ŀ����
�����ȡ��塢��4��Ԫ��ͬ�������ڱ��е�VIIA�壮
��1��4��Ԫ���е縺����С�Ļ�̬ԭ�ӵļ۵����Ų�ʽ�� ��
��2����Ԫ�ؿɹ㷺������������л����У����ȱ�ϩ��CH3-CH=CCl2���DZ�ϩ������̼ԭ�������ӵ�������ԭ�ӱ�������ԭ��ȡ���γɵ�һ���ȴ���������̼ԭ�ӹ�����ӻ������� ���Ȼ�������CrO2Cl2�����л��ϳ��п������������Ȼ��������������л��ﷴӦ��CrO2Cl2������Ϊ���ɫҺ�壬����CCl4��CS2�Ȼ��ܣ��ݴ˿��ж�CrO2Cl2�� ������ԡ��Ǽ��ԡ������ӣ�
��3������ʯ��Na3AlF6����Ҫ�������Al2O3�����������ۼ����侧�岻���磬������ʱ�ܵ��磮�ڱ���ʯ��Na3AlF6�������д��� ����ѡ���
A�����Ӽ� B�����Լ� C����λ�� D�����»���
��4����[��CN��2]��һ�ַ��ӹ��ͺͻ�ѧ������±�ص��ʺ����ƵĻ������Ϊ��±�أ�һ��������к��� ���м���
��1��4��Ԫ���е縺����С�Ļ�̬ԭ�ӵļ۵����Ų�ʽ��
��2����Ԫ�ؿɹ㷺������������л����У����ȱ�ϩ��CH3-CH=CCl2���DZ�ϩ������̼ԭ�������ӵ�������ԭ�ӱ�������ԭ��ȡ���γɵ�һ���ȴ���������̼ԭ�ӹ�����ӻ�������
��3������ʯ��Na3AlF6����Ҫ�������Al2O3�����������ۼ����侧�岻���磬������ʱ�ܵ��磮�ڱ���ʯ��Na3AlF6�������д���
A�����Ӽ� B�����Լ� C����λ�� D�����»���
��4����[��CN��2]��һ�ַ��ӹ��ͺͻ�ѧ������±�ص��ʺ����ƵĻ������Ϊ��±�أ�һ��������к���
���㣺ԭ�Ӻ�������Ų�,�����ijɼ����,���Լ��ͷǼ��Լ�,ԭ�ӹ���ӻ���ʽ���ӻ������ж�
ר�⣺ԭ�������ṹר��,��ѧ���뾧��ṹ
��������1���ǽ���������ΪI����I�ĵ縺����С��I������������Ϊ7��
��2�����ݶ��ȱ�ϩ��CH3-CH=CCl2����Cԭ���γɵĹ��ۼ�������CrO2Cl2������Ϊ���ɫҺ�壬����CCl4��CS2�Ȼ��ܣ������ɷ��ӹ��ɵ����ʣ����������ݿ�֪��Ϊ�Ǽ��Է��ӣ�
��3������ʯ��Na3AlF6�������У��������������������Ӽ���ϣ�Al��F֮����м��Թ��ۼ���Ҳ����λ����
��4����[��CN��2]�Ľṹ��ʽΪN��C-C��N�������к�2���м���
��2�����ݶ��ȱ�ϩ��CH3-CH=CCl2����Cԭ���γɵĹ��ۼ�������CrO2Cl2������Ϊ���ɫҺ�壬����CCl4��CS2�Ȼ��ܣ������ɷ��ӹ��ɵ����ʣ����������ݿ�֪��Ϊ�Ǽ��Է��ӣ�
��3������ʯ��Na3AlF6�������У��������������������Ӽ���ϣ�Al��F֮����м��Թ��ۼ���Ҳ����λ����
��4����[��CN��2]�Ľṹ��ʽΪN��C-C��N�������к�2���м���
���
�⣺��1���ǽ���������ΪI����I�ĵ縺����С��I������������Ϊ7�����Ե�Ļ�̬ԭ�ӵļ۵����Ų�ʽ��5s25p5���ʴ�Ϊ��5s25p5��
��2�����ȱ�ϩ��CH3-CH=CCl2���У�Cû�й¶Ե��ӣ���һ��C�γ�4�����۵���Ϊsp3���ڶ��͵�����C�γ�1��˫����������������۲���Ӷ����ֱ�Ϊ3������sp2��CrO2Cl2������Ϊ���ɫҺ�壬����CCl4��CS2�Ȼ��ܣ������ɷ��ӹ��ɵ����ʣ����������ݿ�֪��Ϊ�Ǽ��Է��ӣ�
�ʴ�Ϊ��sp3��sp2���Ǽ��ԣ�
��3������ʯ��Na3AlF6�������У��������������������Ӽ���ϣ�Al��F֮����м��Թ��ۼ���Ҳ����λ�������Ա���ʯ��Na3AlF6�������д������Ӽ������Լ�����λ�����ʴ�Ϊ��ABC��
��4����[��CN��2]�Ľṹ��ʽΪN��C-C��N�������к�2���м������Թ���2��2=4���м����ʴ�Ϊ��4��
��2�����ȱ�ϩ��CH3-CH=CCl2���У�Cû�й¶Ե��ӣ���һ��C�γ�4�����۵���Ϊsp3���ڶ��͵�����C�γ�1��˫����������������۲���Ӷ����ֱ�Ϊ3������sp2��CrO2Cl2������Ϊ���ɫҺ�壬����CCl4��CS2�Ȼ��ܣ������ɷ��ӹ��ɵ����ʣ����������ݿ�֪��Ϊ�Ǽ��Է��ӣ�
�ʴ�Ϊ��sp3��sp2���Ǽ��ԣ�
��3������ʯ��Na3AlF6�������У��������������������Ӽ���ϣ�Al��F֮����м��Թ��ۼ���Ҳ����λ�������Ա���ʯ��Na3AlF6�������д������Ӽ������Լ�����λ�����ʴ�Ϊ��ABC��
��4����[��CN��2]�Ľṹ��ʽΪN��C-C��N�������к�2���м������Թ���2��2=4���м����ʴ�Ϊ��4��
������������±��Ԫ��Ϊ֪ʶ����������ԭ�ӵĵ����Ų������Ӽ��Լ���ѧ���ȣ�����ԭ�ӽṹ�����ʵĿ��飬��Ŀ�ۺ��Խ�ǿ����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
���й������漰��ѧ�仯��������������ԭ��Ӧ���ǣ�������
| A����ͥ�о�����ʳ������ˮ����ˮ�� |
| B����ɫ��Ӧ�����Ȼ��ƺ��Ȼ�����Һ |
| C���ձ������˵�վ�˷�Ӧ����U-235�ѱ�����-133���-135 |
| D����ͨ�����������ظ���ؼ��˾���Ƿ�ƺ�ݳ� |
���б�ʾ��Ӧ��ѧ��Ӧ�����ӷ���ʽ��ȷ���ǣ�������
| A����������Һ�м��������ˮ��Al3++3OH-=Al��OH��3�� |
| B��NO2��ˮ�ķ�Ӧ��3NO2+H2O=2NO3-+NO+2H+ |
| C���������������Һ��ͨ��������Cl2+2OH-=ClO-+Cl-+H2O |
| D�������ۼ���ϡ�����У�2Fe+6H+=2Fe3++3H2�� |
 ��1����֪25��ʱ��C2H5OH��l����ȼ����Ϊ1366.8kJ/mol�����Ȼ�ѧ����ʽ��ʾ��
��1����֪25��ʱ��C2H5OH��l����ȼ����Ϊ1366.8kJ/mol�����Ȼ�ѧ����ʽ��ʾ��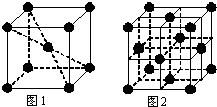 �±��dz�ʽ���ڱ���һ���֣����еı�Ŵ�����Ӧ��Ԫ�أ�
�±��dz�ʽ���ڱ���һ���֣����еı�Ŵ�����Ӧ��Ԫ�أ�