��Ŀ����
(1)��ͼ��ʾ������������ͨ��ʢ�и�����ɫ�����Ĺ��ƿ��ʢ�г�ʪ��ɫ�����Ĺ��ƿ���ɹ۲쵽��������________��

(2)Ϊ��ֹ����β����Ⱦ������������ˮ�����Ե����ʣ�����________��Һ���ն����������ԭ����(�û�ѧ����ʽ��ʾ)________��������һԭ������ҵ�ϳ������۵�ʯ�������չ�ҵ����β���Ƶ�Ư�ۣ�Ư�۵���Ч�ɷ���________(�ѧʽ)������¶���ڿ����е�Ư�ۣ���ϡ����������������________(����ĸ������)��
A��O2
B��Cl2
C��CO2
D��HClO
�𰸣�

��ϰ��ϵ�д�
�����Ŀ
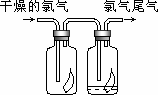
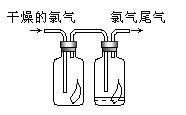

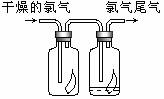 (1)��ͼ��ʾ������������ͨ��ʢ�и�����ɫ�����Ĺ��ƿ��ʢ�г�ʪ��ɫ�����Ĺ��ƿ���ɹ۲쵽��������_________________________��
(1)��ͼ��ʾ������������ͨ��ʢ�и�����ɫ�����Ĺ��ƿ��ʢ�г�ʪ��ɫ�����Ĺ��ƿ���ɹ۲쵽��������_________________________��