��Ŀ����
ijͬѧ��������ͭ����ᾧˮ�����IJⶨʵ�飮���������գ�
��ʵ�鲽�衿
��1���� �����������ƣ���ͬ��ȷ������������������
��2���ڴ������м���Լ2g��ϸ������ͭ���壬��������
��3����ʢ������ͭ����Ĵ������������������������ȣ�ֱ����ɫ��ȫ��ף�Ȼ����������� ����ȴ�����£���������
��4���ظ���3����ʵ����к��ز�����ֱ�����γ������������0.001g��
�����ݼ�¼�봦����
�����ϱ��е����ݴ�����������㱾��ʵ���������Ϊ %����֪x������ֵΪ5����
�����������ۡ�
��5����һ��ʵ�飬������Ҫ���� �Σ������֣���ͬ����������Ҫ���� �Σ�
��6�����ز�����Ŀ���� ��
��7���ظ�����ʵ����xƽ��ֵ��Ŀ���� ��
��8��ʵ��ֵ������ֵƫ���ԭ������� �����ţ���
a�����ȹ������о��彦�� b��������Ʒ�к��м��Ȳ��ӷ�������
c��ʵ��ǰ��������泱ʪ d���������պ�ֱ�ӷ��ڿ�������ȴ��
��ʵ�鲽�衿
��1����
��2���ڴ������м���Լ2g��ϸ������ͭ���壬��������
��3����ʢ������ͭ����Ĵ������������������������ȣ�ֱ����ɫ��ȫ��ף�Ȼ�����������
��4���ظ���3����ʵ����к��ز�����ֱ�����γ������������0.001g��
�����ݼ�¼�봦����
| ��һ��ʵ�� | �ڶ���ʵ�� | |
| ������������g�� | 29.563 | 30.064 |
| ����+������������g�� | 31.676 | 32.051 |
| ���غ�����+����ͭ��������g�� | 30.911 | 31.324 |
| x��ֵ | 5.05 | 5.13 |
�����������ۡ�
��5����һ��ʵ�飬������Ҫ����
��6�����ز�����Ŀ����
��7���ظ�����ʵ����xƽ��ֵ��Ŀ����
��8��ʵ��ֵ������ֵƫ���ԭ�������
a�����ȹ������о��彦�� b��������Ʒ�к��м��Ȳ��ӷ�������
c��ʵ��ǰ��������泱ʪ d���������պ�ֱ�ӷ��ڿ�������ȴ��
���㣺����ͭ�����нᾧˮ�����IJⶨ
ר�⣺ʵ��̽�������ݴ�����
��������1�����ݾ�ȷ��ѡ�����������
��3���ڼ��Ⱥ���ȴʱ��Ϊ�˷�ֹ����ͭ��ˮ��Ӧ������ͭ���ڸ������н�����ȴ���Ӷ��õ��������������ˮ����ͭ��
��4��ȡ����ƽ��ֵ������������
��5��Ϊȷ������ͭ������ȫʧȥ�ᾧˮ���������ȴ�����ټ�����ȴ������ֱ���������γ����IJ����0.1gΪֹ������Ϊ��ȷ����Ʒ�нᾧˮ�Ƿ��Ѿ���ȫ����ȥ��
��6���������γ����������䣬˵��������ȫ��ʧȥ�ᾧˮ��
��7����β���ȡƽ��ֵ�ɼ���żȻ��
��8���ڲⶨ����������Ʒ�к��м��Ȼӷ������ʻ�ʵ��ǰ������Ʒ���泱ʪ��������ɲ������ƫ�ߣ�
��3���ڼ��Ⱥ���ȴʱ��Ϊ�˷�ֹ����ͭ��ˮ��Ӧ������ͭ���ڸ������н�����ȴ���Ӷ��õ��������������ˮ����ͭ��
��4��ȡ����ƽ��ֵ������������
��5��Ϊȷ������ͭ������ȫʧȥ�ᾧˮ���������ȴ�����ټ�����ȴ������ֱ���������γ����IJ����0.1gΪֹ������Ϊ��ȷ����Ʒ�нᾧˮ�Ƿ��Ѿ���ȫ����ȥ��
��6���������γ����������䣬˵��������ȫ��ʧȥ�ᾧˮ��
��7����β���ȡƽ��ֵ�ɼ���żȻ��
��8���ڲⶨ����������Ʒ�к��м��Ȼӷ������ʻ�ʵ��ǰ������Ʒ���泱ʪ��������ɲ������ƫ�ߣ�
���
�⣺��1��������ƽ��ȷ��Ϊ0.1g��������ƽ�ɾ�ȷ��0.001g���������ݿ�֪��Ӧѡ�������ƽ���ʴ�Ϊ��������ƽ��
��3��������ˮ����ͭ���ˮ����Ӧ���ڸ��������ܷ���ȴ���ʴ�Ϊ����������
��4��ʵ���ýᾧˮΪ��
=5.09��ʵ��������=
��100%=1.8%���ʴ�Ϊ��1.8��
��5��Ϊȷ������ͭ������ȫʧȥ�ᾧˮ���������ȴ�����ټ�����ȴ������ֱ���������γ����IJ����0.1gΪֹ������Ϊ��ȷ����Ʒ�нᾧˮ�Ƿ��Ѿ���ȫ����ȥ����������Ҫ����2�Σ�����4�Σ�
�ʴ�Ϊ��2��4��
��6���������γ����������䣬˵��������ȫ��ʧȥ�ᾧˮ���ʴ�Ϊ����֤�ᾧˮȫ��ʧȥ��
��7����β���ȡƽ��ֵ�ɼ���żȻ���ʴ�Ϊ������żȻ��
��8��a�����ȹ��������������彦��������ˮ�������ⶨ���ƫ��a��ȷ��
b��������Ʒ�к��м��Ȳ��ӷ������ʻᵼ�²ⶨ��ˮ������ƫС����b����
c��ʵ��ǰ��������泱ʪ�����Ⱥ�ˮ�ӷ�������ˮ�������ⶨ���ƫ��c��ȷ��
d�����Ⱥ�����δ�������������ȴ���ᵼ�²ⶨ������ͭ������ƫ�ⶨ��ˮ������ƫС����d����
�ʴ�Ϊ��ac��
��3��������ˮ����ͭ���ˮ����Ӧ���ڸ��������ܷ���ȴ���ʴ�Ϊ����������
��4��ʵ���ýᾧˮΪ��
| 5.05+5.13 |
| 2 |
| 5.09-5 |
| 5 |
��5��Ϊȷ������ͭ������ȫʧȥ�ᾧˮ���������ȴ�����ټ�����ȴ������ֱ���������γ����IJ����0.1gΪֹ������Ϊ��ȷ����Ʒ�нᾧˮ�Ƿ��Ѿ���ȫ����ȥ����������Ҫ����2�Σ�����4�Σ�
�ʴ�Ϊ��2��4��
��6���������γ����������䣬˵��������ȫ��ʧȥ�ᾧˮ���ʴ�Ϊ����֤�ᾧˮȫ��ʧȥ��
��7����β���ȡƽ��ֵ�ɼ���żȻ���ʴ�Ϊ������żȻ��
��8��a�����ȹ��������������彦��������ˮ�������ⶨ���ƫ��a��ȷ��
b��������Ʒ�к��м��Ȳ��ӷ������ʻᵼ�²ⶨ��ˮ������ƫС����b����
c��ʵ��ǰ��������泱ʪ�����Ⱥ�ˮ�ӷ�������ˮ�������ⶨ���ƫ��c��ȷ��
d�����Ⱥ�����δ�������������ȴ���ᵼ�²ⶨ������ͭ������ƫ�ⶨ��ˮ������ƫС����d����
�ʴ�Ϊ��ac��
������������һ������ʵ���⣬��Ҫѧ������ʵ��Ĺؼ���ϸ�ڣ���Ҫ���ʵ��������������ԭ����з������ȿ�����ѧ���Ի�ѧʵ��Ļ��������ͳ�����������ʶ��ʹ�ã��ֿ�����ѧ����������ͽ�����������������Ŀ������ѧ����ʵ������Ҫ��ϸߣ�������ʵ���Ͼ���Դ�ڿα���ѧ��ʵ�飬ֻҪѧ�������ȥ���������������˼�������ͻ���ú���Ϥ���ܼ�

��ϰ��ϵ�д�
 ABC����ȫ�ž�ϵ�д�
ABC����ȫ�ž�ϵ�д�
�����Ŀ
��֪W��X��Y��ZΪ������Ԫ�أ����ǵ�ԭ�Ӱ뾶��������W��Z��X��Y�ֱ�ͬ���壬Y��Zͬ���ڣ�Z����X�γ����ֳ��������ӻ�������Ӹ����Ⱦ�Ϊ2��1������˵��������ǣ�������
| A��Y��Z��X��W��ԭ���������μ�С |
| B��W��X ���γɼȺ����Թ��ۼ��ֺ��Ǽ��Թ��ۼ��Ļ����� |
| C����W��X��Y��Z����Ԫ����ɵĻ�����һ�������� |
| D��X��Y�γɵĻ��������������8�����ȶ��ṹ |
���1g SO2�к���n����ԭ�ӣ����ӵ������ǣ�������
| A��32 n mol-1 | ||
| B��64 nmol-1 | ||
C��
| ||
D��
|
Ԫ�صĴ�����̬�У�������
| A�����ʺͻ����� |
| B����Һ����Һ������ |
| C������̬������̬ |
| D��������ͻ���� |
���и߾�������������ֲ�ͬ�ĵ��徭�Ӿ۷�Ӧ�����ɵ��ǣ�������
A��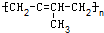 |
B��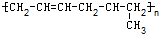 |
C��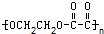 |
D��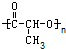 |
��ͬ�¶ȣ������Ϊ0.5L���������������з������淴Ӧ��A��g��+3B��g���T2C��g����H=-92.6kJ?mol-1��ʵ���÷�Ӧ����ʼ���ﵽƽ��ʱ���й��������±���ʾ ������������ȷ���ǣ�������
| ������� | ������������/ | �ﵽƽ��ʱ��ϵ�����仯 | ||
| A | B | C | ||
| �� | 1 | 3 | 0 | ����46.3kJ |
| �� | 0.3 | 0.9 | 1.4 | |
| A����ƽ��ʱ������������C�����ʵ���Ũ�Ⱦ�Ϊ1.0mol?L-1 |
| B�������ٴ�ƽ��Aת������50% |
| C���������д�ƽ��ʱ���յ�����Ϊ18.52kJ |
| D������ʼ������A��B��C��Ϊ0.5mol�����ƽ��ǰ�ԣ��������ԣ��棩 |
����˵����ȷ���ǣ�������
| A��AgClˮ��Һ��Ag+��Cl-Ũ�ȵij˻���һ������ |
| B��AgClˮ��Һ�ĵ����Ժ���������AgClΪ������� |
| C���¶�һ��ʱ����AgClˮ��Һ��Ag+��Cl-Ũ�ȵij˻�����Kspֵʱ������ҺΪAgCl�ı�����Һ |
| D����AgClˮ��Һ�м������ᣬc��Cl-������Kspֵ��� |