��Ŀ����
�±���Ԫ�����ڱ���һ���֣���Ա��еĢ١�����Ԫ�أ���Ԫ�ط��Ż�ѧʽ��ջش��������⣺
��1������ЩԪ���У���������ǿ��Ԫ���� ������õķǽ���Ԫ���� ����̬�⻯�����ȶ������ʵĵ���ʽΪ ��
��2����ѧ��������õ�Ԫ���� ����ԭ�ӵ�ԭ�ӽṹʾ��ͼΪ ����Ԫ���γɵ��ʵĽṹʽΪ ��
��3��Ԫ�ص�����������Ӧ��ˮ������������ǿ���� ��������ǿ���� �������Ե����������� ��д������֮�����Ӧ�����ӷ���ʽ �� �� ��
��4���ڢۡ���Ԫ���У�ԭ�Ӱ뾶������ ��ԭ�Ӱ뾶��С���� ��
��5���ڢ����ĵ����У���ѧ���ʽϻ��õ��� ���û�ѧ��Ӧ˵������ʵ��д����Ӧ�Ļ�ѧ����ʽ���� ��
��6���õ���ʽ��ʾ�������ɻ�������γɹ��� ��
| ���� ���� | ��A | ��A | ��A | ��A | VA | ��A | ��A | 0 |
| �� | �� | �� | ||||||
| �� | �� | �� | �� | �� | �� | �� | ||
| �� | �� | �� |
��2����ѧ��������õ�Ԫ����
��3��Ԫ�ص�����������Ӧ��ˮ������������ǿ����
��4���ڢۡ���Ԫ���У�ԭ�Ӱ뾶������
��5���ڢ����ĵ����У���ѧ���ʽϻ��õ���
��6���õ���ʽ��ʾ�������ɻ�������γɹ���
���㣺Ԫ�������ɺ�Ԫ�����ڱ����ۺ�Ӧ��
ר�⣺Ԫ����������Ԫ�����ڱ�ר��
��������Ԫ�������ڱ���λ�ã���֪��ΪC����ΪF����ΪNa����ΪMg����ΪAl����ΪSi����ΪCl����ΪAr����ΪK����ΪBr��
��1��ͬ����������ҽ����Լ������ǽ�������ǿ��ͬ�������϶��½�������ǿ���ǽ����Լ���������Ԫ����F�ķǽ�������ǿ��HF���ȶ���������Hԭ����Fԭ��֮���γ�1�Թ��õ��Ӷԣ�
��2��ϡ������Ar��ѧ��������ã�Arԭ�Ӻ�����18�����Ӳ㣬��3�����Ӳ㣬���������Ϊ2��8��8��
��Ԫ���γɵ���ΪN2��������Nԭ��֮���γ����Թ��õ��Ӷԣ�
��3����Ԫ��û����ۺ����ᣬ������������Ӧ��ˮ������������ǿ���Ǹ����ᣬ����Ԫ����K�Ľ�������ǿ�����������صļ�����ǿ�����������������������
���������������ط�Ӧ���ɸ��������ˮ��������������Ӧ���ɸ���������ˮ�������������������Ʒ�Ӧ����ƫ��������ˮ��
��4��ͬ�����������ԭ�Ӱ뾶��С��
��5��ͬ�������϶��·ǽ����Լ����������õ���֮�����û���Ӧ˵����
��6���������ɻ�����ΪMgCl2����þ�����������ӹ��ɣ���Mgԭ�ӡ�Clԭ�ӵ���ʽ��ʾ���γɣ�
��1��ͬ����������ҽ����Լ������ǽ�������ǿ��ͬ�������϶��½�������ǿ���ǽ����Լ���������Ԫ����F�ķǽ�������ǿ��HF���ȶ���������Hԭ����Fԭ��֮���γ�1�Թ��õ��Ӷԣ�
��2��ϡ������Ar��ѧ��������ã�Arԭ�Ӻ�����18�����Ӳ㣬��3�����Ӳ㣬���������Ϊ2��8��8��
��Ԫ���γɵ���ΪN2��������Nԭ��֮���γ����Թ��õ��Ӷԣ�
��3����Ԫ��û����ۺ����ᣬ������������Ӧ��ˮ������������ǿ���Ǹ����ᣬ����Ԫ����K�Ľ�������ǿ�����������صļ�����ǿ�����������������������
���������������ط�Ӧ���ɸ��������ˮ��������������Ӧ���ɸ���������ˮ�������������������Ʒ�Ӧ����ƫ��������ˮ��
��4��ͬ�����������ԭ�Ӱ뾶��С��
��5��ͬ�������϶��·ǽ����Լ����������õ���֮�����û���Ӧ˵����
��6���������ɻ�����ΪMgCl2����þ�����������ӹ��ɣ���Mgԭ�ӡ�Clԭ�ӵ���ʽ��ʾ���γɣ�
���
�⣺��Ԫ�������ڱ���λ�ã���֪��ΪC����ΪF����ΪNa����ΪMg����ΪAl����ΪSi����ΪCl����ΪAr����ΪK����ΪBr��
��1��ͬ����������ҽ����Լ������ǽ�������ǿ��ͬ�������϶��½�������ǿ���ǽ����Լ���������Ԫ����K�Ľ�������ǿ��F�ķǽ�������ǿ��HF���ȶ���������Hԭ����Fԭ��֮���γ�1�Թ��õ��Ӷԣ������ʽΪ ���ʴ�Ϊ��K��F��
���ʴ�Ϊ��K��F�� ��
��
��2��ϡ������Ar��ѧ��������ã�Arԭ�Ӻ�����18�����Ӳ㣬��3�����Ӳ㣬���������Ϊ2��8��8�������ʾ��ͼΪ�� ��
��
��Ԫ���γɵ���ΪN2��������Nԭ��֮���γ����Թ��õ��Ӷԣ��ṹʽΪN��N��
�ʴ�Ϊ��Ar�� ��N��N��
��N��N��
��3��FԪ�صķǽ�������ǿ����FԪ��û������������ˮ�������������ˮ����������ǿ����HClO4������Ԫ����K�Ľ�������ǿ����KOH�ļ�����ǿ��Al��OH��3�������������������������֮�����ӷ�Ӧ����ʽΪ�ֱ�Ϊ��H++OH-=H2O��Al��OH��3+3H+=Al3++3H2O��Al��OH��3+OH-=AlO2-+2H2O��
�ʴ�Ϊ��HClO4��KOH��Al��OH��3��H++OH-=H2O��Al��OH��3+3H+=Al3++3H2O��Al��OH��3+OH-=AlO2-+2H2O��
��4��ͬ�����������ԭ�Ӱ뾶��С���ۡ���Ԫ���У�ԭ�Ӱ뾶������Na��ԭ�Ӱ뾶��С����Cl���ʴ�Ϊ��Na��Cl��
��5��ͬ�������϶��·ǽ����Լ������ʻ�����Cl2��Br2�������õ���֮�����û���Ӧ˵������ѧ��Ӧ����ʽΪ��Cl2+2NaBr=2NaCl+Br2���ʴ�Ϊ��Cl2��Cl2+2NaBr=2NaCl+Br2��
��6���������ɻ�����ΪMgCl2����þ�����������ӹ��ɣ���Mgԭ�ӡ�Clԭ�ӵ���ʽ��ʾ���γɣ� ��
��
�ʴ�Ϊ�� ��
��
��1��ͬ����������ҽ����Լ������ǽ�������ǿ��ͬ�������϶��½�������ǿ���ǽ����Լ���������Ԫ����K�Ľ�������ǿ��F�ķǽ�������ǿ��HF���ȶ���������Hԭ����Fԭ��֮���γ�1�Թ��õ��Ӷԣ������ʽΪ
 ���ʴ�Ϊ��K��F��
���ʴ�Ϊ��K��F�� ��
����2��ϡ������Ar��ѧ��������ã�Arԭ�Ӻ�����18�����Ӳ㣬��3�����Ӳ㣬���������Ϊ2��8��8�������ʾ��ͼΪ��
 ��
����Ԫ���γɵ���ΪN2��������Nԭ��֮���γ����Թ��õ��Ӷԣ��ṹʽΪN��N��
�ʴ�Ϊ��Ar��
 ��N��N��
��N��N����3��FԪ�صķǽ�������ǿ����FԪ��û������������ˮ�������������ˮ����������ǿ����HClO4������Ԫ����K�Ľ�������ǿ����KOH�ļ�����ǿ��Al��OH��3�������������������������֮�����ӷ�Ӧ����ʽΪ�ֱ�Ϊ��H++OH-=H2O��Al��OH��3+3H+=Al3++3H2O��Al��OH��3+OH-=AlO2-+2H2O��
�ʴ�Ϊ��HClO4��KOH��Al��OH��3��H++OH-=H2O��Al��OH��3+3H+=Al3++3H2O��Al��OH��3+OH-=AlO2-+2H2O��
��4��ͬ�����������ԭ�Ӱ뾶��С���ۡ���Ԫ���У�ԭ�Ӱ뾶������Na��ԭ�Ӱ뾶��С����Cl���ʴ�Ϊ��Na��Cl��
��5��ͬ�������϶��·ǽ����Լ������ʻ�����Cl2��Br2�������õ���֮�����û���Ӧ˵������ѧ��Ӧ����ʽΪ��Cl2+2NaBr=2NaCl+Br2���ʴ�Ϊ��Cl2��Cl2+2NaBr=2NaCl+Br2��
��6���������ɻ�����ΪMgCl2����þ�����������ӹ��ɣ���Mgԭ�ӡ�Clԭ�ӵ���ʽ��ʾ���γɣ�
 ��
���ʴ�Ϊ��
 ��
��
���������⿼��Ԫ�����ڱ���Ԫ�������ɣ��Ѷ��еȣ����ض�Ԫ���������뻯ѧ����Ŀ��飬ע�������õ���ʽ��ʾ��ѧ�������ʵ��γɣ����ս����ԡ��ǽ�����ǿ���Ƚϼ�ʵ����ʵ��

��ϰ��ϵ�д�
�����Ŀ
��50ml0.01mol/LH2SO4��Һ����50ml0.01mol/LNaOH��Һʱ��������Һ�ʣ�������
| A�����ж� | B������ |
| C������ | D������ |
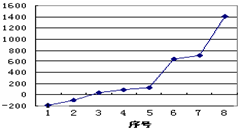 ��1����������8��Ԫ�ذ������۵�ߵ͵�˳��
��1����������8��Ԫ�ذ������۵�ߵ͵�˳��
 �±���Ԫ�����ڱ���һ���֣��������е���ĸ�ֱ����һ�ֻ�ѧԪ�أ�
�±���Ԫ�����ڱ���һ���֣��������е���ĸ�ֱ����һ�ֻ�ѧԪ�أ�