��Ŀ����
(����Ԥ����)(1)���ڵ��۵⻯����Һ�У��μ������������Ƽ�����Һ�������ῴ����Һ����ɫ��������Ϊ________�����ӷ���ʽΪ__________________________��
���ڵ�͵����γɵ���ɫ��Һ�У��μ��������Ƽ�����Һ��������ɫ����ʧ��������Ϊ______________________________�����ӷ���ʽ��_______________________________��
���Ա�������ʵ�����õĽ������I2��ClO����SO42������ǿ������˳������Ϊ_____________________________��
(2)������Ƭ��ͭƬ�����ʵ��֤��������ʵ����д����Ӧ�Ļ�ѧ����ʽ��
��Ũ����������Ա�ϡ����ǿ��________________________________��
���Ȼ�����Һ��Fe3���������Ա�����ͭ��Һ�е�Cu2��ǿ��__________________________��
�����Ļ�ԭ�Ա�ͭǿ��
___________________________________________________________��
(1)��NaClO��KI����������I2
ClO����2I����H2O=I2��Cl����2OH��
��I2��Na2SO3��ԭ������
SO32����2OH��=SO42������H2O
��ClO����I2��SO42��
(2)��Cu��Ũ���ᷴӦ����ϡ�����Ӧ��
Cu��2H2SO4(Ũ)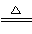 ��SO2����2H2O
��SO2����2H2O
��Cu����FeCl3��Һ��Ӧ��
2FeCl3��Cu=2FeCl2��CuCl2
��Fe����CuSO4��Һ��Ӧ�û���Cu��
Fe��CuSO4=FeSO4��Cu(�𰸲�Ψһ����������)
��������(1)���õ�����I2������������֤NaClO�������Ժ�Na2SO3�Ļ�ԭ�ԣ���д��ѧ����ʽʱע����Һ����Ϊ���ԡ�(2)�оٳ�����Ӧ��ʵ���ɡ�







