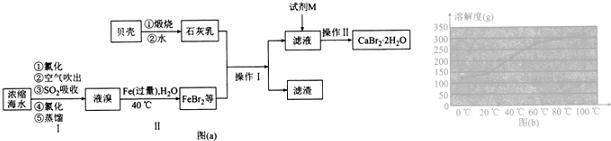
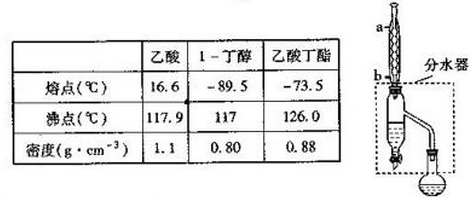
��Ŀ����
13������18.4mol/L��ŨH2SO4������500mL0.2mol/L��ϡH2SO4���ɹ�ѡ��������У��ٲ���������ƿ���ձ��ܽ�ͷ�ιܢ���Ͳ��������ƽ��ҩ�ף���ش��������⣺
��1�����������У�������ϡH2SO4ʱ����Ҫʹ�õ��Тڢޢߣ�����ţ�����ȱ�ٵ�������500mL����ƿ��
��2���������Ƶ�ϡH2SO4���вⶨ������Ũ�ȴ���0.2mol/L������������ƹ����п�������Ũ��ƫ�ߵ�ԭ������㣩��Һ��δ��ȴ���̶��ݡ�����ʱ���ӿ̶��ߣ�
���� ��1������������Һ��һ�㲽��ѡ����Ҫ��������
��2������C=$\frac{n}{V}$��֪������ʹnƫ�������VƫС�IJ�������ʹ��ҺŨ��ƫ�ߣ�
��� �⣺��1����Ũ��������һ�����ʵ���Ũ��ϡ����һ�㲽�裺���㡢��ȡ��ϡ�͡���ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȵȣ��õ�����������Ͳ����ͷ�ιܡ����������ձ�������ƿ������500mL0.2mol/L��ϡ����Ӧѡ��500mL����ƿ�������ò�������������ƿ��������ƽ��ҩ�ף�����Ҫ��������500mL����ƿ��
�ʴ�Ϊ���ڢޢߣ�500mL����ƿ��
��2������C=$\frac{n}{V}$��֪������ʹnƫ�������VƫС�IJ�������ʹ��ҺŨ��ƫ�ߣ�������Һ��δ��ȴ���̶��ݣ�������Һ���ƫС������ʱ���ӿ̶��ߣ�������Һ���ƫС��
�ʴ�Ϊ����Һ��δ��ȴ���̶��ݣ�����ʱ���ӿ̶��ߣ�
���� ���⿼����һ�����ʵ���Ũ����Һ�����ƣ���ȷ����ԭ�������������ǽ���ؼ���ע��������������ʵ����ʵ�������Һ�����Ӱ�죬����C=$\frac{n}{V}$�����������ķ�������Ŀ�ѶȲ���

��ϰ��ϵ�д�
 ��ѧ����ϵ�д�
��ѧ����ϵ�д�
�����Ŀ
8����ij�¶��£���2molA��3molB�����ܱ������з�����Ӧ��aA��g��+B��g���TC��g��+D��g����5min��ﵽƽ�⣨��֪�÷�Ӧ�Ļ�ѧƽ�ⳣ��Ϊ1���������¶Ȳ��䣮�������������10�˱���A ��ת���ʲ��䣮��B��ת����Ϊ��������
| A�� | 60% | B�� | 40% | C�� | 24% | D�� | 4% |
3����������к���Cr��OH��3��Al2O3��ZnO��CuO��NiO�����ʣ���ҵ��ͨ���������̻���Na2Cr2O7�����ʣ�
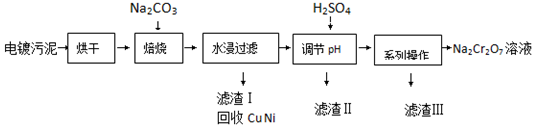
��֪��
��Cr��OH��3��ZnO��Al2O3һ����Ϊ���Ի����
��Na2Cr2O7��Na2SO4�ڲ�ͬ�¶��µ��ܽ�������
��1�����չ���������Na2CrO4�Ļ�ѧ����ʽΪ4Cr��OH��3+4Na2CO3+3O2$\frac{\underline{\;����\;}}{\;}$4Na2CrO4+6H2O+4CO2��ˮ������Һ�г�Na2CrO4�����ڵ�������NaAlO2��Na2ZnO2��
��2������H2SO4����pH��Ŀ��Ϊ��ȥ��Һ��NaAlO2��Na2ZnO2���ʣ�����CrO42-ת��ΪCr2O72-��
��3���õ�������ġ�ϵ�в������IJ���Ϊ����Ũ������ȴ�ᾧ ���ˣ�
��4����1L����H2SO4��������Һ�к���Ԫ������Ϊ23.4g��CrO42-��8/9ת��ΪCr2O72-����ת����������Һ��c��Cr2O72-��=0.2mol•L-1��
��5����Na2Cr2O7��H2SO4���Һ�м���H2O2���ټ������Ѳ�ҡ�������Ѳ�Ϊ��ɫ������������CrO5��������Ӧ�Ļ�ѧ����ʽΪNa2Cr2O7+4H2O2+H2SO4�T2CrO5+Na2SO4+5H2O��

��֪��
��Cr��OH��3��ZnO��Al2O3һ����Ϊ���Ի����
��Na2Cr2O7��Na2SO4�ڲ�ͬ�¶��µ��ܽ�������
| 20�� | 60�� | 100�� | |
| Na2SO4 | 19.5 | 45.3 | 42.5 |
| Na2Cr2O7 | 183 | 269 | 415 |
��2������H2SO4����pH��Ŀ��Ϊ��ȥ��Һ��NaAlO2��Na2ZnO2���ʣ�����CrO42-ת��ΪCr2O72-��
��3���õ�������ġ�ϵ�в������IJ���Ϊ����Ũ������ȴ�ᾧ ���ˣ�
��4����1L����H2SO4��������Һ�к���Ԫ������Ϊ23.4g��CrO42-��8/9ת��ΪCr2O72-����ת����������Һ��c��Cr2O72-��=0.2mol•L-1��
��5����Na2Cr2O7��H2SO4���Һ�м���H2O2���ټ������Ѳ�ҡ�������Ѳ�Ϊ��ɫ������������CrO5��������Ӧ�Ļ�ѧ����ʽΪNa2Cr2O7+4H2O2+H2SO4�T2CrO5+Na2SO4+5H2O��