��Ŀ����
A��B��C��D��EΪԭ���������������5�ֶ�����Ԫ�أ�F��һ�ֺ��ص�������Ϊ56��������Ϊ30��A��ͬλ������һ�ֺ��������ӣ�B������ϵ������Ǵ�����������������CԪ������⻯��Y��ˮ��Һ�ʼ��ԣ�E�Ƕ������е縺����С��Ԫ�أ�D�ɷֱ���A��B��C��E����Ԫ���γ�ԭ�Ӹ����Ȳ�ͬ�ij���������ش��������⣺
��1��д��A��E��Ԫ���γɵ�ԭ�Ӹ�����Ϊ1��1�Ļ�����ĵ���ʽ ��
��2��B��C��D�ĵ�һ�������ɴ�С����Ϊ����Ԫ�ط��ţ��� ��
��3��Y������ԭ�ӵ��ӻ��������Ϊ �����ӵ����幹��Ϊ ��
��4����A��B��D��E�γɵĻ�����W��Z������W������Ԫ����ɣ�Z������Ԫ����ɣ�ˮ��Һ���Լ��ԣ������ӷ���ʽ��ʾZ��Һ�Լ��Ե�ԭ�� ����Ũ�ȣ��������W��Z�Ļ����Һ�У�����Ũ���ɴ�С��˳��Ϊ ��
��5��F�ĺ�������Ų�ʽΪ�� ��F��һ�ֺ������FO42-����ǿ�����ԣ����������м���ϡ���ᣬ��Һ��Ϊ��ɫ��������ɫ��ζ����������÷�Ӧ�����ӷ���ʽ�� ��
��6��BԪ�ص�ij���⻯��Ϊֱ���ͷ��ӣ�������1g����̬�⻯����ȫȼ�������ȶ�������ʱ�ų�����50kJ�����ʾ���⻯��ȼ���ȵ��Ȼ�ѧ����ʽΪ�� ��
��1��д��A��E��Ԫ���γɵ�ԭ�Ӹ�����Ϊ1��1�Ļ�����ĵ���ʽ
��2��B��C��D�ĵ�һ�������ɴ�С����Ϊ����Ԫ�ط��ţ���
��3��Y������ԭ�ӵ��ӻ��������Ϊ
��4����A��B��D��E�γɵĻ�����W��Z������W������Ԫ����ɣ�Z������Ԫ����ɣ�ˮ��Һ���Լ��ԣ������ӷ���ʽ��ʾZ��Һ�Լ��Ե�ԭ��
��5��F�ĺ�������Ų�ʽΪ��
��6��BԪ�ص�ij���⻯��Ϊֱ���ͷ��ӣ�������1g����̬�⻯����ȫȼ�������ȶ�������ʱ�ų�����50kJ�����ʾ���⻯��ȼ���ȵ��Ȼ�ѧ����ʽΪ��
���㣺λ�ýṹ���ʵ����ϵӦ��
ר�⣺
������A��B��C��D��EΪԭ���������������5�ֶ�����Ԫ�أ�A��ͬλ������һ�ֺ��������ӣ���AΪHԪ�أ�
B������ϵ������Ǵ�����������������ԭ��ֻ����2�����Ӳ㣬����������Ϊ4����BΪ̼Ԫ�أ�
CԪ������⻯��Y��ˮ��Һ�ʼ��ԣ�YΪ��������CΪNԪ�أ�
E�Ƕ������е縺����С��Ԫ�أ���EΪNaԪ�أ�
D�����A��B��C��E����Ԫ�طֱ��γ�ԭ�Ӹ����Ȳ���ͬ�ij������������֪DΪOԪ�أ�
F��һ�ֺ��ص�������Ϊ56��������Ϊ30��F��������Ϊ26����FΪFeԪ�أ��ݴ˽��н��
B������ϵ������Ǵ�����������������ԭ��ֻ����2�����Ӳ㣬����������Ϊ4����BΪ̼Ԫ�أ�
CԪ������⻯��Y��ˮ��Һ�ʼ��ԣ�YΪ��������CΪNԪ�أ�
E�Ƕ������е縺����С��Ԫ�أ���EΪNaԪ�أ�
D�����A��B��C��E����Ԫ�طֱ��γ�ԭ�Ӹ����Ȳ���ͬ�ij������������֪DΪOԪ�أ�
F��һ�ֺ��ص�������Ϊ56��������Ϊ30��F��������Ϊ26����FΪFeԪ�أ��ݴ˽��н��
���
�⣺A��B��C��D��EΪԭ���������������5�ֶ�����Ԫ�أ�A��ͬλ������һ�ֺ��������ӣ���AΪHԪ�أ�B������ϵ������Ǵ�����������������ԭ��ֻ����2�����Ӳ㣬����������Ϊ4����BΪ̼Ԫ�أ�CԪ������⻯��Y��ˮ��Һ�ʼ��ԣ�YΪ��������CΪNԪ�أ�E�Ƕ������е縺����С��Ԫ�أ���EΪNaԪ�أ�D�����A��B��C��E����Ԫ�طֱ��γ�ԭ�Ӹ����Ȳ���ͬ�ij������������֪DΪOԪ�أ�F��һ�ֺ��ص�������Ϊ56��������Ϊ30��F��������Ϊ26����FΪFeԪ�أ�
��1��H��Na��Ԫ���γɵ�ԭ�Ӹ�����Ϊ1��1�Ļ�����ΪNaH���������ӻ���������������⸺���ӹ��ɣ������ʽΪ�� ��
��
�ʴ�Ϊ�� ��
��
��2��ͬһ����Ԫ�صĵ�һ����������ԭ������������������ڢ�A��Ĵ��ڵڢ�A��ģ����ڵ�ԭ���ǰ����ṹ���ȶ����������һ�����ܴ�С˳���ǣ�N��O��C��
�ʴ�Ϊ��N��O��C��
��3��YΪNH3������Nԭ�ӳ�3���Ҽ�����һ�Թµ��Ӷԣ��ӻ������ĿΪ4��Nԭ�Ӳ�ȡsp3�ӻ������ӵ����幹��Ϊ�����Σ�
�ʴ�Ϊ��sp3�������Σ�
��4����H��C��O��Na�γɵĻ�����W��Z������W������Ԫ����ɣ�Z������Ԫ����ɣ�ˮ��Һ���Լ��ԣ�W��Z������Һ�й��棬��WΪNa2CO3��ZΪNaHCO3��NaHCO3��Һ��HCO3-ˮ�⣺HCO3-+H2O?H2CO3+OH-���ƻ�ˮ�ĵ���ƽ�⣬��Һ�ʼ��ԣ�
��Ũ�ȡ��������NaHCO3��Na2CO3�Ļ����Һ�У���Һ��CO32-��HCO3-������ˮ�⣬��HCO3-��ˮ����ڵ���̶ȣ���Һ�ʼ��ԣ���c��OH-����c��H+��������Һ��CO32-ˮ��̶ȴ���HCO3-����c��HCO3-����c��CO32-������Һ��c��Na+�����ˮ��̶�����������Ũ���ɴ�С��˳��Ϊ��c��Na+����c��HCO3-����c��CO32-����c��OH-����c��H+����
�ʴ�Ϊ��HCO3-+H2O?H2CO3+OH-��c��Na+����c��HCO3-����c��CO32-����c��OH-����c��H+����
��5��FΪFeԪ�أ���ԭ�ӵĺ����������Ϊ26�����ݹ���ԭ������ԭ�Ӻ�������Ų�ʽΪ��1s22s22p63s23p63d64s2��
Fe��һ�ֺ������FeO42-����ǿ�����ԣ����������м���ϡ���ᣬ��Һ��Ϊ��ɫ��������ɫ��ζ���������˵����Ӧ�����������Ӻ��������÷�Ӧ�����ӷ���ʽΪ��4FeO42-+20H+�T4Fe3++3O2��+10H2O��
�ʴ�Ϊ��1s22s22p63s23p63d64s2�� 4FeO42-+20H+�T4Fe3++3O2��+10H2O��
��6��BΪCԪ�أ�CԪ�ص�ij���⻯��Ϊֱ���ͷ��ӣ�������Ϊ��Ȳ��������1g����̬�⻯����ȫȼ�������ȶ�������ʱ�ų�����50kJ��1mol��Ȳ������Ϊ26g���ų�������Ϊ��50kJ��26=1300kJ�����ʾ���⻯��ȼ���ȵ��Ȼ�ѧ����ʽΪ��C2H2��g��+
O2��g��=2CO2��g��+H2O��l����H=-1300 kJ?mol-1��
�ʴ�Ϊ��C2H2��g��+
O2��g��=2CO2��g��+H2O��l����H=-1300 kJ?mol-1��
��1��H��Na��Ԫ���γɵ�ԭ�Ӹ�����Ϊ1��1�Ļ�����ΪNaH���������ӻ���������������⸺���ӹ��ɣ������ʽΪ��
 ��
���ʴ�Ϊ��
 ��
����2��ͬһ����Ԫ�صĵ�һ����������ԭ������������������ڢ�A��Ĵ��ڵڢ�A��ģ����ڵ�ԭ���ǰ����ṹ���ȶ����������һ�����ܴ�С˳���ǣ�N��O��C��
�ʴ�Ϊ��N��O��C��
��3��YΪNH3������Nԭ�ӳ�3���Ҽ�����һ�Թµ��Ӷԣ��ӻ������ĿΪ4��Nԭ�Ӳ�ȡsp3�ӻ������ӵ����幹��Ϊ�����Σ�
�ʴ�Ϊ��sp3�������Σ�
��4����H��C��O��Na�γɵĻ�����W��Z������W������Ԫ����ɣ�Z������Ԫ����ɣ�ˮ��Һ���Լ��ԣ�W��Z������Һ�й��棬��WΪNa2CO3��ZΪNaHCO3��NaHCO3��Һ��HCO3-ˮ�⣺HCO3-+H2O?H2CO3+OH-���ƻ�ˮ�ĵ���ƽ�⣬��Һ�ʼ��ԣ�
��Ũ�ȡ��������NaHCO3��Na2CO3�Ļ����Һ�У���Һ��CO32-��HCO3-������ˮ�⣬��HCO3-��ˮ����ڵ���̶ȣ���Һ�ʼ��ԣ���c��OH-����c��H+��������Һ��CO32-ˮ��̶ȴ���HCO3-����c��HCO3-����c��CO32-������Һ��c��Na+�����ˮ��̶�����������Ũ���ɴ�С��˳��Ϊ��c��Na+����c��HCO3-����c��CO32-����c��OH-����c��H+����
�ʴ�Ϊ��HCO3-+H2O?H2CO3+OH-��c��Na+����c��HCO3-����c��CO32-����c��OH-����c��H+����
��5��FΪFeԪ�أ���ԭ�ӵĺ����������Ϊ26�����ݹ���ԭ������ԭ�Ӻ�������Ų�ʽΪ��1s22s22p63s23p63d64s2��
Fe��һ�ֺ������FeO42-����ǿ�����ԣ����������м���ϡ���ᣬ��Һ��Ϊ��ɫ��������ɫ��ζ���������˵����Ӧ�����������Ӻ��������÷�Ӧ�����ӷ���ʽΪ��4FeO42-+20H+�T4Fe3++3O2��+10H2O��
�ʴ�Ϊ��1s22s22p63s23p63d64s2�� 4FeO42-+20H+�T4Fe3++3O2��+10H2O��
��6��BΪCԪ�أ�CԪ�ص�ij���⻯��Ϊֱ���ͷ��ӣ�������Ϊ��Ȳ��������1g����̬�⻯����ȫȼ�������ȶ�������ʱ�ų�����50kJ��1mol��Ȳ������Ϊ26g���ų�������Ϊ��50kJ��26=1300kJ�����ʾ���⻯��ȼ���ȵ��Ȼ�ѧ����ʽΪ��C2H2��g��+
| 5 |
| 2 |
�ʴ�Ϊ��C2H2��g��+
| 5 |
| 2 |
���������⿼����λ�á��ṹ�����ʹ�ϵ���ۺ�Ӧ�ã���Ŀ�Ѷ��еȣ������漰��һ�����ܴ�С�Ƚϡ��Ȼ�ѧ����ʽ��д������Ũ�ȴ�С�Ƚϡ�����ʽ��д��֪ʶ������֪ʶ��϶࣬�ۺ��Խ�ǿ����ֿ�����ѧ�����Ӧ�û���֪ʶ��������

��ϰ��ϵ�д�
�����Ŀ
��NA��ʾ�����ӵ�����������˵����ȷ���ǣ�������
| A��1L0.1mol?L-1�İ�ˮ�к��е�NH3?H2O������Ϊ0.1NA |
| B��2.3 g�������������������Ӧ�����ۼ������ת�Ƶ�������Ϊ0.1NA |
| C�����Ե缫���ʳ��ˮ������·��ͨ��1 NA���ӣ���������������11.2L |
| D����⾫��ͭʱ����������������6.4g�����·��ת�Ƶ�����Ϊ0.2NA |
���������п϶�A������B�����Ļ�����ǿ���ǣ�������
| A��Aԭ�ӵ�������������Bԭ�ӵ������������� |
| B��A����������Ϊ���Ի����B����������Ϊ�� |
| C��AԪ�صĵ縺�Ա�BԪ�صĵ縺��С |
| D��1Ħ��A�������û���H+���ɵ�������1Ħ��B�������û���H+���ɵ������� |
 �ס�����Ԫ�������ڱ��е����λ�����±����������ԭ���������3�����һ�ֵ�������Ȼ��Ӳ���������ʣ���������ͬ����Ԫ�أ������ж���ȷ���ǣ�������
�ס�����Ԫ�������ڱ��е����λ�����±����������ԭ���������3�����һ�ֵ�������Ȼ��Ӳ���������ʣ���������ͬ����Ԫ�أ������ж���ȷ���ǣ�������| A�������ԣ��ף��ң��� |
| B��ԭ�Ӱ뾶�������죾�� |
| C���������Ԫ��ԭ�Ӻ�����������13 |
| D���γɵ�����⻯����ȶ��ԣ������� |
��500g��1.17g NaCl��0.84g NaF����Һ�У��μӹ�����AgNO3��Һ�����ã����ˣ�ϴ�ӣ����صõ�2.87g���壬�ɴ˿��Եó�����ȷ�����ǣ�������
| A��������ֻ��һ���ֲμӷ�Ӧ |
| B��������ֻ��һ���ֲμӷ�Ӧ |
| C����������AgNO3����Һ���������� |
| D��������������ˮ |
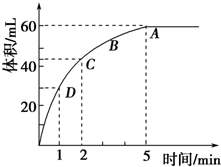 Ϊ���о�MnO2��˫��ˮ��H2O2���ķ�Ӧ���ʣ�ijѧ������������MnO2 ��ĩ�� 50mL�ܶ�Ϊ1.1g?cm-3 ��˫��ˮ��Һ�У�ͨ��ʵ��ⶨ���ڱ�״���·ų�����������ʱ��Ĺ�ϵ����ͼ��ʾ������ͼ�ش��������⣺
Ϊ���о�MnO2��˫��ˮ��H2O2���ķ�Ӧ���ʣ�ijѧ������������MnO2 ��ĩ�� 50mL�ܶ�Ϊ1.1g?cm-3 ��˫��ˮ��Һ�У�ͨ��ʵ��ⶨ���ڱ�״���·ų�����������ʱ��Ĺ�ϵ����ͼ��ʾ������ͼ�ش��������⣺