��Ŀ����
A~J��Ϊ�л����������֮���ת������ͼ��ʾ��

ʵ�������
��D���ܷ���������Ӧ������������Ʒ�Ӧ�ų�������
�ں˴Ź������ױ���F�������������⣬��������֮��Ϊ1��1��1��
��G��ʹ������Ȼ�̼��Һ��ɫ��
��1 mol J�����������Ʒ�Ӧ�ɷų�22. 4 L��������״������
�����������Ϣ�ش��������⣺
��1��A�Ľṹ��ʽΪ______________�������������칹������A����B�ķ�Ӧ������_________��Ӧ��
��2��D�Ľṹ��ʽΪ__________________��
��3����E����F�Ļ�ѧ����ʽΪ____________________________��E�еĹ�������____________�������ƣ�����E������ͬ�����ŵ�E��ͬ���칹�廹��__________________��д���ṹ��ʽ�������������칹����
��4��G�Ľṹ��ʽΪ__________________��
��5����I����J�Ļ�ѧ����ʽΪ_________________________��
��D���ܷ���������Ӧ������������Ʒ�Ӧ�ų�������
�ں˴Ź������ױ���F�������������⣬��������֮��Ϊ1��1��1��
��G��ʹ������Ȼ�̼��Һ��ɫ��
��1 mol J�����������Ʒ�Ӧ�ɷų�22. 4 L��������״������
�����������Ϣ�ش��������⣺
��1��A�Ľṹ��ʽΪ______________�������������칹������A����B�ķ�Ӧ������_________��Ӧ��
��2��D�Ľṹ��ʽΪ__________________��
��3����E����F�Ļ�ѧ����ʽΪ____________________________��E�еĹ�������____________�������ƣ�����E������ͬ�����ŵ�E��ͬ���칹�廹��__________________��д���ṹ��ʽ�������������칹����
��4��G�Ľṹ��ʽΪ__________________��
��5����I����J�Ļ�ѧ����ʽΪ_________________________��
��1��BrCH2CH=CHCH2Br��ȡ������ˮ�⣩
��2��
��3��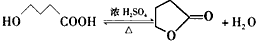 ���Ȼ����ǻ���
���Ȼ����ǻ���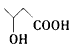 ��
�� ��
��
 ��
��
��4��
��5��

��3��
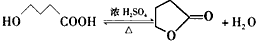 ���Ȼ����ǻ���
���Ȼ����ǻ���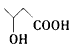 ��
�� ��
�� ��
��
��4��

��5��


��ϰ��ϵ�д�
�����Ŀ
|
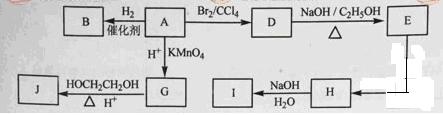 ��ͼ��A-J��Ϊ�л����������ͼ�е���Ϣ���ش��������⣺
��ͼ��A-J��Ϊ�л����������ͼ�е���Ϣ���ش��������⣺��1����״������A����Է�������Ϊ82�����к�̼87.80%������12.2%��B��һ�ȴ������һ�֣�B�Ľṹ��ʽΪ ��3�֣�
��2��M��B��һ��ͬ���칹�壬M��ʹ������Ȼ�̼��Һ��ɫ�����������е�̼ԭ�ӹ�ƽ�棬��M�Ľṹ��ʽΪ ��3�֣���
��3����
 A����D�ķ�Ӧ������ ��3�֣�����D����E�ķ�Ӧ������ ��3�֣���
A����D�ķ�Ӧ������ ��3�֣�����D����E�ķ�Ӧ������ ��3�֣�����4��G�ķ���ʽΪC6H10O4, 0.146gG����20ml0.100mol/L NaOH��Һ��ȫ�кͣ�J��һ�ָ߷��ӻ��������Gת��ΪJ�Ļ�ѧ����ʽΪ ��4�֣���
��5��H�к��й������� ��3�֣��� I�к��еĹ������� ��3�֣���


