��Ŀ����
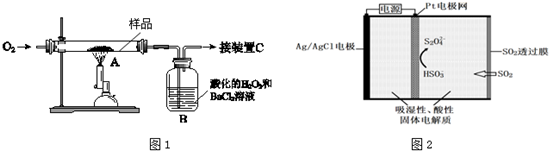
11��ij��ҩ��Ҫ������������FeS2����ijѧϰС������ͼ1��ʾװ�ý���ʵ�飬�ⶨ��������Ԫ�ص�������������1��װ���У�AΪ��������װ�ã�BΪ��������װ�ã�CΪ���β�����Ƿ���SO2�Ĵ�������ȡm g����ҩ��Ʒ��װ��A�У�����ַ�Ӧ��ʹ��Ԫ��ȫ��ת��ΪSO2��SO3����B�еõ���ɫ��������������װ��C��δ��SO2��
��װ��B�е�H2O2��Ӧʱ���ֳ��������ԣ�
����������Ԫ�ص������������������������B�г�����������
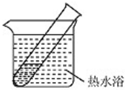
�۴������Ĺ���ԭ����ͼ2��ʾ��
�������������ĵ缫��Ӧʽ��2HSO3-+2H++2e-=S2O42-+2H2O��
��2��Ϊ�ⶨ��Ԫ�ص���������������ʵ�飮
a����Aװ���е�ʣ��������������������������H2��������ַ�Ӧ����ˣ��õ���ɫ��Һ��
b������Һ�еμ�TiCl3��Һ����ǡ����ȫ��Ӧ��TiCl3������ΪTiO2+��
c���õζ����ⶨFe2+����������v mL n mol/LK2Cr2O7��Һ��
��a����Һ���еĽ�����������Fe3+��Fe2+��
��b�з�Ӧ�����ӷ���ʽ��Fe3++Ti3++H2O=Fe2++TiO2++2H+��
��c��K2Cr2O7����ԭΪCr3+����Ʒ����Ԫ��������������ѧ��ʾʽ��$\frac{33.6nv}{m}%$��
���� ��1����ȡm g����ҩ��Ʒ��װ��A�У�����ַ�Ӧ��FeS2ȼ�����ɶ������������������������ͨ���ữ�Ĺ���������Ȼ�����Һ�У�ʹ��Ԫ��ȫ��ת��ΪSO2��SO3����B�еõ���ɫ����������������ԭ��Ӧ�������ᱵ������
��Ԫ���غ��������Ҫ�ⶨB�г�������������Ԫ�����ʵ�����
������������ԭ��Ӧ������Һ��������������ӵõ���������S2O42-��
��2���ٽ�Aװ���е�ʣ��������������������������H2��������ַ�Ӧ����ˣ��õ���ɫ��Һ��˵������������������
������Һ�еμ�TiCl3��Һ����ǡ����ȫ��Ӧ��TiCl3������������ΪTiO2+��
���õζ����ⶨFe2+����������v mL n mol/LK2Cr2O7��Һ�����ݻ�ѧ��Ӧ�Ķ�����ϵ����õ���
��� �⣺��1����ȡm g����ҩ��Ʒ��װ��A�У�����ַ�Ӧ��FeS2ȼ�����ɶ������������������������ͨ���ữ�Ĺ���������Ȼ�����Һ�У�ʹ��Ԫ��ȫ��ת��ΪSO2��SO3����B�еõ���ɫ����������������ԭ��Ӧ�������ᱵ�����������������Ҫ����������������+4����Ϊ+6����������ӣ�װ��B�е�H2O2��Ӧʱ���ֳ��������ԣ�
�ʴ�Ϊ��������
����������Ԫ�ص�������������Ҫ�ⶨB�г�������������Ԫ�����ʵ��������Բ�����������B�г�����������
�ʴ�Ϊ��B�г�����������
������������ԭ��Ӧ������Һ��������������ӵõ���������S2O42-���缫��ӦΪ2HSO3-+2H++2e-=S2O42-+2H2O��
�ʴ�Ϊ��2HSO3-+2H++2e-=S2O42-+2H2O��
��2���ٽ�Aװ���е�ʣ��������������������������H2����֤������������ַ�Ӧ����ˣ��õ���ɫ��Һ��˵�����������������������ܽ��õ���Һ�к���������Ϊ��Fe3+��Fe2+��
�ʴ�Ϊ��Fe3+��Fe2+��
������Һ�еμ�TiCl3��Һ����ǡ����ȫ��Ӧ��TiCl3������������ΪTiO2+����Ӧ�����ӷ���ʽΪ��Fe3++Ti3++H2O=Fe2++TiO2++2H+��
�ʴ�Ϊ��Fe3++Ti3++H2O=Fe2++TiO2++2H+��
���õζ����ⶨFe2+����������v mL n mol/LK2Cr2O7��Һ����
6Fe2++Cr2O72-+14H+=6Fe3++2Cr3++7H2O
6 1
n��Fe2+�� v��10-3L��n mol/L
n=6vn��10-3mol
��Ʒ����Ԫ����������=$\frac{6vn��1{0}^{-3}mol��56g/mol}{mg}$��100%=$\frac{0.336}{m}$vn��100%=$\frac{0.336vn}{m}$��100%=$\frac{33.6nv}{m}%$��
�ʴ�Ϊ��$\frac{33.6nv}{m}%$��
���� ���⿼����������ɵķ����жϣ���Ҫ��ʵ�鹤����������Ӧʵ�ʵ�����Ӧ�ã��������ʺͷ�Ӧ���������Ӧ�ã����ջ����ǹؼ�����Ŀ�Ѷ��еȣ�

| A�� | 0.1mol•L-1��NaHS��Һ������Ũ�ȹ�ϵ��c��OH-��=c��H+��-c��S2-��+c��H2S�� | |
| B�� | Ũ��Ϊ0.1mol•L-1��̼������Һ��c��Na+��=2c��CO32-��+c��HCO3-��+c��H2CO3�� | |
| C�� | pH=12�İ�ˮ��pH=2������������ϣ�c��Cl-����c��NH4+����c��H+����c��OH-�� | |
| D�� | ������Һ��NaOH��Һ���Ϻ�������Һ�����ԣ�c��Na+����c��CH3COO-�� |
| A�� | ij��ɫ������Һ�У�Ca2+��NH4+��CO32-��HCOO- | |
| B�� | �ܽ���AlCl3����Һ�У�Na+��K+��SO42-��S2- | |
| C�� | 25��ʱ��ˮ�������c��H+��=1��l0-l3mol/L����Һ�У�K+��Ba2+��NO3-��I- | |
| D�� | ����Ũ�Ⱦ�Ϊ0.1mol/L����Һ�У�Na+��Fe3+��CH3COO-��NO3- |
| װ�� | ʵ����� | �Թ��е�ҩƷ | ���� |
 | ʵ��� | 2mL������Һ���� �ν�ŨNaOH��Һ | �����ݲ����� һ��ʱ�����Һ ��ڣ��Թܱ� �������� |
| ʵ��� | 2mL������Һ�� ����Ũ��ˮ | �����ݲ����� һ��ʱ�����Һ �����Ա仯 |
a��Ag��NH3��2++2H2O=Ag++2NH3+H2O b��AgOH���ȶ������ֽ�Ϊ��ɫAg2O
��1������������Һ�����ҩƷ��AgNO3��Һ��Ũ��ˮ��
��2�������飬ʵ������������NH3����ɫ��������Ag2O��
����ʪ��ĺ�ɫʯ����ֽ����NH3����������������ֽ������
�ڲ���Ag2O��ԭ������NaOH�����£����ȴٽ�NH3•H2O�ֽ⣬�ݳ�NH3����ʹƽ��Ag��NH3��2++2H2O?Ag++2NH3•H2O�����ƶ���c��Ag+������Ag+��OH-��Ӧ2OH-+2Ag+=Ag2O+H2O������ת��ΪAg2O��
��3����ͬѧ�Բ���������ԭ��������裺������NaOH��ԭAg2O��ʵ�鼰������AgNO3��Һ�м������NaOH��Һ�����ֺ�ɫ������ˮԡ���ȣ�δ����������
��4�����¼��裺��NaOH�����£�������NH3����ԭAg2O����ͼ1��ʾװ�ý���ʵ�飮�����������������߿��ڻ�������ʯ�Һ�Ũ��ˮ��ȡNH3��װ�ü�ͼ���г������ԣ���

��5����ͬѧ��Ϊ�ڣ�4����ʵ���л���Ag��NH3��2OH���ɣ��ɴ���������裺��NaOH�����£�������Ag
��NH3��2OHҲ������NH3��ԭAg2O�ķ�Ӧ��������ͼ2��ʵ�飺
���в���Ag2O�ܽ��ڰ�ˮ�У��÷�Ӧ�Ļ�ѧ����ʽ��Ag2O+4 NH3•H2O=2Ag��NH3��2OH+3H2O��
��ʵ����֤ʵ������������ݵ�����������Һ�Ӵ����Թܱ�������������
��6����HNO3��ϴ�Թܱ��ϵ�Ag���÷�Ӧ�Ļ�ѧ����ʽ��4HNO3��ϡ��+3Ag�T3AgNO3+NO��+2H2O��
| A�� |  Al��OH��3 | B�� |  C6H12O6 | C�� |  CH3CH2OH | D�� |  NaHCO3 |
 ��
��