��Ŀ����
 �������������ɫ�ͳ����µ�Ksp���±���ʾ��
�������������ɫ�ͳ����µ�Ksp���±���ʾ��| Cu��OH��2 | CuOH | CuCl | Cu2O | |
| ��ɫ | ��ɫ | ��ɫ | ��ɫ | ש��ɫ |
| Ksp��25�棩 | 1.6��10-19 | 1.0��10-14 | 1.2��10-6 | - |
��1���������е������ӷ�Ӧ����ʽΪ
��2��Ϊ��ȷ����Դ���������������в���һ����֮��Ч����
A���۲����������������ɫ
B����U�ι����˷ֱ�������η�̪��Һ
C����ȼ�ŵ�ľ������U�ιܿ�
D����U�ιܿ���һ��ʪ��ĵ���KI��ֽ
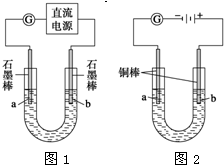
ʵ�����������װ�õ�ʯī������ͭ������ֱ����Դ���е�⣬װ����ͼ2��ʾ��
�۲쵽������������ʾ��
�ٿ�ʼ�������������Һ�����µ�ͭ�������䰵��
��5min��b��������ʼ���ְ�ɫ�������������࣬����a����ɢ��
��10min�����a���İ�ɫ������ʼ��ɺ�ɫ��
��12min��b�������İ�ɫ������ʼ��ɻ�ɫ��Ȼ����ɳȻ�ɫ��
��a��һֱ�д������ݲ�����
��ֹͣ��⣬��U�ι�������Һ����һ��ʱ����ϲ���Һ����ɫ��û�г�����ɫ���²����ȫ����ש��ɫ��
��3��a�������ĵ缫��Ӧ����ʽΪ
��4�����5min��b�������ĵ缫��Ӧ����ʽΪ
��5��12min��b���������ֵijȻ�ɫ�����ijɷ���
���㣺���ԭ��
ר�⣺�绯ѧר��
��������1����ⱥ��ʳ��ˮ�õ��IJ������������ơ�������������
��2���ڵ���Ȼ��Ƶ�װ���У��͵�Դ�������������������ü��ϻ�����������������������������ü��ϲ��������������ƺ����������ݲ�����������жϵ缫��
��3���ڵ��ص��������������ӷ����õ��ӵĻ�ԭ��Ӧ��
��4���������⣺���5min��������ɫ�������ش�
��5�����ݱ������ʵ���ɫ��CuOH�ǻ�ɫ�������䲻�ȶ��ֽ�Ϊש��ɫ��������ͭ���ش��жϣ�
��2���ڵ���Ȼ��Ƶ�װ���У��͵�Դ�������������������ü��ϻ�����������������������������ü��ϲ��������������ƺ����������ݲ�����������жϵ缫��
��3���ڵ��ص��������������ӷ����õ��ӵĻ�ԭ��Ӧ��
��4���������⣺���5min��������ɫ�������ش�
��5�����ݱ������ʵ���ɫ��CuOH�ǻ�ɫ�������䲻�ȶ��ֽ�Ϊש��ɫ��������ͭ���ش��жϣ�
���
�⣺��1����ⱥ��ʳ��ˮ�õ��IJ������������ơ��������������ʴ�Ϊ��2Cl-+2H2O
2OH-+H2��+Cl2�����ʴ�Ϊ��2Cl-+2H2O
2OH-+H2��+Cl2����
��2������Ȼ��Ƶ�װ���У��͵�Դ�������������������ü��ϻ�����������������������������ü��ϲ��������������ƺ�������
A�����ص������Ͽ��Բ�������������ɫ�����壬�����Բ�����������ɫ���壬�ʹ۲����������������ɫ�������ж�����������A����
B����U�ι����˷ֱ�������η�̪��Һ�����ɫ�ļ�����������Ӧ���ǵ�Դ�ĸ�������B��ȷ��
C����ȼ�ŵ�ľ������U�ιܿڣ�ֻ�ܼ���������������������ʱ�ĵ缫��Ӧ�����Ƕ��������ʵIJ��������飬��C����
D����U�ιܿ���һ��ʪ��ĵ���KI��ֽ����ʹʪ��ĵ���KI��ֽ����֤�������IJ������õ缫����������Ӧ���ǵ�Դ����������D��ȷ��
��ѡBD��
��3��a�����������ڵ��ص���������ˮ�е������������ӷ����õ��ӵĻ�ԭ��Ӧ��2H++2e-=H2������2H2O+2e-�T2OH-+H2������
�ʴ�Ϊ��2H++2e-=H2������2H2O+2e-�T2OH-+H2������
��4����������֪�����5min��b�������İ�ɫ�������ӱ���֪������������CuCl�����Ըü��Ϸ����ĵ缫��Ӧ����ʽΪCu+Cl--e-�TCuCl����
�ʴ�Ϊ��Cu+Cl--e-�TCuCl����
��5�����������ϱ������ʵ���ɫ֪����12min��b���������ֵijȻ�ɫ�����ijɷ���CuOH��Cu2O����ΪKsp��CuOH����Ksp��CuCl�������Ե�������CuClת��Ϊ��ɫ��CuOH������CuOH���ȶ��ֽ�����Cu2O���������ʵ���ɫΪ��ɫ��
�ʴ�Ϊ��CuOH��Cu2O��Ksp��CuOH����Ksp��CuCl����CuClת��Ϊ��ɫ��CuOH������CuOH���ȶ��ֽ�����Cu2O�����ԳȻ�ɫ�����ijɷ�ΪCuOH��Cu2O�Ļ���
| ||
| ||
��2������Ȼ��Ƶ�װ���У��͵�Դ�������������������ü��ϻ�����������������������������ü��ϲ��������������ƺ�������
A�����ص������Ͽ��Բ�������������ɫ�����壬�����Բ�����������ɫ���壬�ʹ۲����������������ɫ�������ж�����������A����
B����U�ι����˷ֱ�������η�̪��Һ�����ɫ�ļ�����������Ӧ���ǵ�Դ�ĸ�������B��ȷ��
C����ȼ�ŵ�ľ������U�ιܿڣ�ֻ�ܼ���������������������ʱ�ĵ缫��Ӧ�����Ƕ��������ʵIJ��������飬��C����
D����U�ιܿ���һ��ʪ��ĵ���KI��ֽ����ʹʪ��ĵ���KI��ֽ����֤�������IJ������õ缫����������Ӧ���ǵ�Դ����������D��ȷ��
��ѡBD��
��3��a�����������ڵ��ص���������ˮ�е������������ӷ����õ��ӵĻ�ԭ��Ӧ��2H++2e-=H2������2H2O+2e-�T2OH-+H2������
�ʴ�Ϊ��2H++2e-=H2������2H2O+2e-�T2OH-+H2������
��4����������֪�����5min��b�������İ�ɫ�������ӱ���֪������������CuCl�����Ըü��Ϸ����ĵ缫��Ӧ����ʽΪCu+Cl--e-�TCuCl����
�ʴ�Ϊ��Cu+Cl--e-�TCuCl����
��5�����������ϱ������ʵ���ɫ֪����12min��b���������ֵijȻ�ɫ�����ijɷ���CuOH��Cu2O����ΪKsp��CuOH����Ksp��CuCl�������Ե�������CuClת��Ϊ��ɫ��CuOH������CuOH���ȶ��ֽ�����Cu2O���������ʵ���ɫΪ��ɫ��
�ʴ�Ϊ��CuOH��Cu2O��Ksp��CuOH����Ksp��CuCl����CuClת��Ϊ��ɫ��CuOH������CuOH���ȶ��ֽ�����Cu2O�����ԳȻ�ɫ�����ijɷ�ΪCuOH��Cu2O�Ļ���
������������һ���йص��صĹ���ԭ�����ۺϿ����⣬Ҫ��ѧ�����з����ͽ��������������ѶȽϴ�

��ϰ��ϵ�д�
 ���ɿ��õ�Ԫ����AB��ϵ�д�
���ɿ��õ�Ԫ����AB��ϵ�д� С�����ϵ�д�
С�����ϵ�д�
�����Ŀ
���º����£���ѧ��Ӧ2NO2��g��?N2O4��g�� �ﵽƽ�������������ͨ��һ���������N2O4�����������������
| A������ | B������ |
| C����С | D�����ж� |
Ϊ��ȥij���������������ʣ���ѡ�õ��Լ������������ȷ���ǣ�������
| ��� | ���� | ���� | ������Ӧѡ�õ��Լ���������� |
| �� | KNO3��Һ | KOH | ����ϡHNO3ͬʱ��pH��ֽ�ⶨ����Һ������ |
| �� | FeSO4��Һ | CuSO4 | ����������۳�ַ�Ӧ������ |
| �� | H2 | CO2 | ����ͨ��ʢ��NaOH��Һ��Ũ�����ϴ��ƿ |
| �� | BaSO4 | BaCO3 | �������ܽ⡢���ˡ�ϴ�ӡ����� |
| A���٢ڢ� | B���ڢۢ� |
| C���٢ۢ� | D���٢ڢۢ� |
�������ӿ���ͬһ��Һ�д������棬���γ���ɫ��Һ��һ���ǣ�������
| A��Ag+��Na+��OH-��Cl- |
| B��Al3+��Fe2+��NO3-��Cl- |
| C��Mg2+��Na+��OH-��SO42- |
| D��K+��Ba2+��NO3-��OH- |