��Ŀ����
 2009��12���ڸ籾�����ٿ������Ϲ�����仯�����,���������������ŷš���
2009��12���ڸ籾�����ٿ������Ϲ�����仯�����,���������������ŷš���
Ϊ�������۵Ľ�������,��ν��ʹ�����CO2�ĺ�������Ч�ؿ�������CO2�������˸�������
�����ӡ�
��Ŀǰ��ҵ����һ�ַ�������CO2������ȼ�ϼ״���Ϊ̽
����Ӧԭ�����ֽ�������ʵ�飬�����Ϊ1 L���ܱ������У�
����3mol CO2��8mol H2��һ�������·�����Ӧ��
CO2(g)+3H2(g)  CH3OH(g)+H2O(g)
��H=��49.0kJ/mol
CH3OH(g)+H2O(g)
��H=��49.0kJ/mol
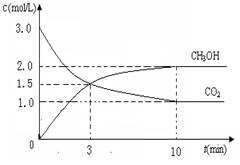
���CO2��CH3OH(g)��Ũ����ʱ��仯��ͼ��ʾ��
�ٴӷ�Ӧ��ʼ��ƽ�⣬������ƽ����Ӧ����v(H2)��___________mol/(L��min)
��CO2��ת����Ϊ ��������һλС����
��ƽ�ⳣ��KֵΪ_____________���������¶�Kֵ��________��
�����д�ʩ����ʹn(CH3OH)��n(CO2)�������___________��
A�������¶� B������He(g)��ʹ��ϵѹǿ����
C����H2O(g)����ϵ�з��� D���ٳ���1mol CO2��3mol H2
�����������������Ʒ�Ӧ2CO��2C��O2����H��0����S��0��������CO����Ⱦ������
�ж��Ƿ���в�˵�����ɣ�______________________________________________��

