��Ŀ����
��6�֣�ij��ɫ����Һ�п��ܴ�������OH-��Cl-��Na+��Mg2+��Cu2+�еļ������ӡ�
��1�������κ�ʵ��Ϳ��Կ϶�ԭ��Һ�в����ڵ�������_______��
��2��ȡ����ԭ��Һ��������������ų���˵��ԭ��Һ�п϶����ڵ�������___________���йص����ӷ���ʽΪ��____________________________��
��3����ȡ����ԭ��Һ�������ϡ���ᣬ�ټӼ���AgNO3��Һ�����ְ�ɫ������˵��ԭ��Һ�п϶����ڵ�������_________��
��4��ԭ��Һ��_________����϶����ڡ� �����϶������ڡ��� ����ȷ������ Na+��
��6�֣� (1) Cu2+ ��(2) OH- 2Al+2OH-+2H2O=2Al3++3H2�� ��(3) Cl- ��(4) �϶����ڣ�ÿ��1�֣����ӷ���ʽ2�֣�
��������
�����������1������ɫ����Һ�в����ܴ���������ɫ��Cu2+����2������Al�����Ի������Һ�ж��ᷴӦ��������������ȡ����ԭ��Һ��������������ų���˵��ԭ��Һ�п϶����ڵ�������OH-���йص����ӷ���ʽΪ2Al+2OH-+2H2O=2Al3++3H2������3����ȡ����ԭ��Һ�������ϡ���ᣬ�ټӼ���AgNO3��Һ�����ְ�ɫ������˵��ԭ��Һ�п϶����ڵ�������Cl-�������ķ�Ӧ��Cl-+Ag+=AgCl����(4)������������ԭ��Һ�к���OH-��Cl-�����Ƕ��������ӣ�����ݵ���غ㣬��Һ��һ�����������ӣ���Cu2+�����ڣ�Mg2+��OH-���ܴ�������Ҳ���ܴ��ڣ�����һ���Ậ��Na+��
���㣺������Һ�����ӵĴ��ڡ����ӹ��漰���ӷ���ʽ����д��֪ʶ��

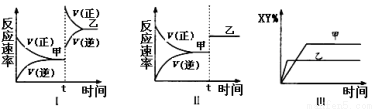
 2XY��g������H<0���ﵽ��ƽ�⡣�ڽ��ı�ijһ�����ﵽ��ƽ�⣬���з�����ȷ����
2XY��g������H<0���ﵽ��ƽ�⡣�ڽ��ı�ijһ�����ﵽ��ƽ�⣬���з�����ȷ����