��Ŀ����
���ܵĴ洢������Ӧ�õ���Ҫƿ������λ�⻯��������廯������Ŀǰ�����õ���Ҫ������ϣ�
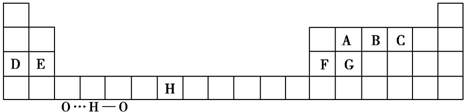
��1��Ti��BH4��2��һ�ֹ���Ԫ�����⻯�ﴢ����ϣ��ڻ�̬Ti2+�У�����ռ�ݵ�����ܲ����Ϊ �����ܲ���е�ԭ�ӹ����Ϊ ��
��2��Һ���Ǹ������ʣ������ܵ��������壬����N2+3H2?2NH3ʵ�ִ�������⣮����˵����ȷ���� ��
a��NH3�����е�ԭ�ӵĹ���ӻ���ʽΪsp2�ӻ�
b��NH+4��PH+4��CH4��BH-4��ClO-4��Ϊ�ȵ�����
c����ͬѹǿʱ��NH3�ķе��PH3�ķе��
d��[Cu��NH3��4]2+�����У�Nԭ������λԭ��
��3����֪NF3��NH3�Ŀռ乹����ͬ����NF3������Cu2+�γ������ӣ���ԭ���� ��
��1��Ti��BH4��2��һ�ֹ���Ԫ�����⻯�ﴢ����ϣ��ڻ�̬Ti2+�У�����ռ�ݵ�����ܲ����Ϊ
��2��Һ���Ǹ������ʣ������ܵ��������壬����N2+3H2?2NH3ʵ�ִ�������⣮����˵����ȷ����
a��NH3�����е�ԭ�ӵĹ���ӻ���ʽΪsp2�ӻ�
b��NH+4��PH+4��CH4��BH-4��ClO-4��Ϊ�ȵ�����
c����ͬѹǿʱ��NH3�ķе��PH3�ķе��
d��[Cu��NH3��4]2+�����У�Nԭ������λԭ��
��3����֪NF3��NH3�Ŀռ乹����ͬ����NF3������Cu2+�γ������ӣ���ԭ����
���㣺�����ijɼ����,ԭ�Ӻ�������Ų�,���ȵ���ԭ������Ӧ��,ԭ�ӹ���ӻ���ʽ���ӻ������ж�
ר�⣺
��������1��д��Ti2+��������Ų�ʽ���ж�������ܲ���ܲ��µ�ԭ�ӹ������
��2��a�����ݼ۲���ӶԻ�������ȷ���ӻ���ʽ��
b���ȵ�����Ϊԭ������Ⱥͼ۵�������ȵ�ԭ���ţ�
c�����Ӽ����������۷е�ߣ�
d���ṩ�µ��ӶԵ�ԭ������ԭ�ӣ�
��3�����������ṹ�жϣ�Cu2+�ṩ�չ����NH3��NF3������ԭ��Nԭ���ṩ�µ��Ӷԣ�����NH3��NF3�й��õ��ӶԵ�ƫת�жϣ�
��2��a�����ݼ۲���ӶԻ�������ȷ���ӻ���ʽ��
b���ȵ�����Ϊԭ������Ⱥͼ۵�������ȵ�ԭ���ţ�
c�����Ӽ����������۷е�ߣ�
d���ṩ�µ��ӶԵ�ԭ������ԭ�ӣ�
��3�����������ṹ�жϣ�Cu2+�ṩ�չ����NH3��NF3������ԭ��Nԭ���ṩ�µ��Ӷԣ�����NH3��NF3�й��õ��ӶԵ�ƫת�жϣ�
���
�⣺��1����̬Ti2+�۵����Ų�ʽΪ��3d2������ܲ�ΪM�㣬���ܲ��º���1��3s�����3��3p�����5��3d���������9��ԭ�ӹ����
�ʴ�Ϊ��M��9��
��2��a��NH3������Nԭ�Ӻ���3�����õ��ӶԺ�һ���µ��Ӷԣ�������۲���Ӷ���4������sp3�ӻ�����a����
b���ȵ�����Ϊԭ������Ⱥͼ۵�������ȵ�ԭ���ţ�NH+4��PH+4��CH4��BH-4������5��ԭ���ţ��Ҽ۵��Ӿ�Ϊ8��Ϊ�ȵ����壬��ClO-4�۵�����Ϊ32�������ڵȵ����壬��b����
c�����Ӽ����������۷е�ߣ���ͬѹǿʱ�������к��������PH3�в������������NH3�е��PH3�ߣ���c��ȷ��
d���ṩ�µ��ӶԵ�ԭ������ԭ�ӣ�[Cu��NH3��4]2+�����У�Nԭ���ṩ�¶Ե��ӣ�Nԭ��Ϊ��ԭ�ӣ���d��ȷ��
�ʴ�Ϊ��cd��
��3�����������ṹ�жϣ�Cu2+�ṩ�չ����NH3��NF3������ԭ��Nԭ���ṩ�µ��Ӷԣ�����N��F��H����Ԫ�صĵ縺��ΪF��N��H����NF3�У����õ��Ӷ�ƫ��F��ƫ��Nԭ�ӣ�ʹ�õ�ԭ���ϵŵ��Ӷ�������Cu2+�γ���λ����
�ʴ�Ϊ��N��F��H����Ԫ�صĵ縺��ΪF��N��H����NF3�У����õ��Ӷ�ƫ��F��ƫ��Nԭ�ӣ�ʹ�õ�ԭ���ϵŵ��Ӷ�������Cu2+�γ���λ����
�ʴ�Ϊ��M��9��
��2��a��NH3������Nԭ�Ӻ���3�����õ��ӶԺ�һ���µ��Ӷԣ�������۲���Ӷ���4������sp3�ӻ�����a����
b���ȵ�����Ϊԭ������Ⱥͼ۵�������ȵ�ԭ���ţ�NH+4��PH+4��CH4��BH-4������5��ԭ���ţ��Ҽ۵��Ӿ�Ϊ8��Ϊ�ȵ����壬��ClO-4�۵�����Ϊ32�������ڵȵ����壬��b����
c�����Ӽ����������۷е�ߣ���ͬѹǿʱ�������к��������PH3�в������������NH3�е��PH3�ߣ���c��ȷ��
d���ṩ�µ��ӶԵ�ԭ������ԭ�ӣ�[Cu��NH3��4]2+�����У�Nԭ���ṩ�¶Ե��ӣ�Nԭ��Ϊ��ԭ�ӣ���d��ȷ��
�ʴ�Ϊ��cd��
��3�����������ṹ�жϣ�Cu2+�ṩ�չ����NH3��NF3������ԭ��Nԭ���ṩ�µ��Ӷԣ�����N��F��H����Ԫ�صĵ縺��ΪF��N��H����NF3�У����õ��Ӷ�ƫ��F��ƫ��Nԭ�ӣ�ʹ�õ�ԭ���ϵŵ��Ӷ�������Cu2+�γ���λ����
�ʴ�Ϊ��N��F��H����Ԫ�صĵ縺��ΪF��N��H����NF3�У����õ��Ӷ�ƫ��F��ƫ��Nԭ�ӣ�ʹ�õ�ԭ���ϵŵ��Ӷ�������Cu2+�γ���λ����
���������⿼�����ʽṹ�����ʣ��漰��������Ų����ӻ���ʽ���жϵ�֪ʶ�㣬ע��[Cu��NH3��4]2+���ӵ�����ԭ�����ṩ�չ����ԭ�ӣ��Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
����ijЩ���ӵļ��鼰����һ����ȷ���ǣ�������
A������ϡ���������ɫ���壬������ͨ�����ʯ��ˮ�У���Һ����ǣ�һ����CO
| ||
B�������Ȼ�����Һ�а�ɫ�����������ټ����ᣬ��������ʧ��һ����SO
| ||
C����������������Һ�����ȣ�������������ʹʪ���ɫʯ����ֽ��������һ����NH
| ||
| D�������Һ�м���KSCN��Һ����������ˮ���ΪѪ��ɫ�������Һ�к���Fe2+ |
һ�������£����ڿ��淴ӦX��g��+3Y��g��?2Z��g������X��Y��Z����ʼŨ�ȷֱ�Ϊc1��c2��c3������Ϊ�㣩���ﵽƽ��ʱ��X��Y��Z��Ũ�ȷֱ�Ϊ0.1mol/L��0.3mol/L��0.08mol/L���������жϲ���ȷ���ǣ�������
| A��c1��c2=1��3 |
| B���÷�Ӧһ���dz�����Ӧ����ƽ�� |
| C��X��Y��ת������� |
| D��c1��ȡֵ��ΧΪ0��c1��0.14 mol/L |
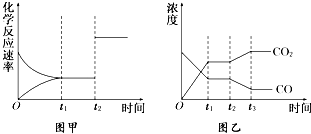 ��֪��ѧ��Ӧ�٣�Fe��s��+CO2��g��?FeO��s��+CO��g������ƽ�ⳣ��ΪK1��
��֪��ѧ��Ӧ�٣�Fe��s��+CO2��g��?FeO��s��+CO��g������ƽ�ⳣ��ΪK1��