��Ŀ����
18��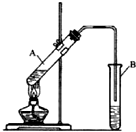 ʵ������ȡ���������ķ�Ӧװ����ͼ��ʾ����ش��������⣮
ʵ������ȡ���������ķ�Ӧװ����ͼ��ʾ����ش��������⣮��1���Թ�A��ʢ�ŵ�ҩƷ�Т�Ũ��������������ᣬ������ҩƷ�����˳���Ǣڢ� �ۣ�����ţ���
��2���Թ�B��ʢ�ŵ�Һ���DZ���̼������Һ����������BCD��������ĸ����
A���кͻӷ�������ζ�� C���кͻӷ�����������
B�����ջӷ����������� D�����������������ܽ�ȣ����ڷֲ�
��3���Թ�B�еĵ��ܲ�����Һ���µ�ԭ���Ƿ�������
��4������һ��ʱ����ֲ쵽�Թ�B��Һ����ϲ� ��ѡ��ϲ㡱���¡�������ɫ��״Һ�����ɣ���Ӧ�Ļ�ѧ����ʽΪCH3COOH+CH3CH2OH$?_{��}^{Ũ����}$CH3COOCH2CH3+H2O��
��Ҫ�����Թ�B�е��������������õ�����Ҫ���������������Ƿ�Һ©����
��5��ͨ����ʵ�黹�ܵó��й������������ǿ���Ľ�������������Ա�̼��ǿ��
���� ��1�������ӷ����Ҵ����ܶȱ�Ũ�����С�����Ũ�����ϡ�ͽ��н��
��2������̼������Һ�����ᷴӦ��ȥ���ᡢͬʱ���������������ܽ�ȣ����ڷֲ㣻
��3����������Һ���¿��ܷ���������
��4�������������ܶ�С��1��������ˮ��������Ӧ�ı���Ϊ�����ǻ��������⣬�÷�Ӧ��������������ˮ����Ϊ���淴Ӧ�����뻥�����ܵ�Һ�壬���÷�Һ�ķ������룻
��5��̼���ƺ����ᷴӦ���ɶ�����̼��
��� �⣺��1��Ũ�����ϡ���ǽ�Ũ�������ˮ�У��ӱ߽��裬Ǩ�Ƶ��˴��������ڴ��Թ��м����Ҵ���Ȼ������������ע�����ᣬ�����Ͻ��裬�����װ���Ҵ���Ũ����Ļ����Ĵ��Թ��м������ᣬ�Լ�����˳��Ϊ���ڢ٢ۣ�
�ʴ�Ϊ���ڢ٣�
��2���Ʊ���������ʱ���ñ���̼������Һ��Ŀ�����кͻӷ����������ᣬʹ֮ת��Ϊ����������ˮ�У�������������������ζ���ܽ�ӷ��������Ҵ�����������������ˮ�е��ܽ�ȣ����ڷֲ�õ�����ѡBCD��
�ʴ�Ϊ������̼������Һ��BCD��
��3�����ܲ��ܲ�����Һ�У�����Ҫ���ڱ���̼������Һ��Һ���ϣ�����Һ���¿��ܷ����������ʴ�Ϊ����������
��4���Թ��м��뱥��Na2CO3��Һ������������Ա�̼��ǿ�����������ܺ�̼���Ʒ�Ӧ���ɶ�����̼���壬��������������ˮ�����ܶȱ�ˮС�����ϲ㣬ʵ�������Ҵ����������÷���������Ӧ��������Ӧ�ı���Ϊ�����ǻ��������⣬�������Ҵ���Ũ���������¼��ȷ���������Ӧ��������������ˮ���÷�ӦΪ���淴Ӧ����ѧ����ʽΪ��CH3COOH+CH3CH2OH$?_{��}^{Ũ����}$CH3COOCH2CH3+H2O��������������ʱ�Ƚ�ʢ�л������Թܳ�����ñ���̼������Һ�кͻӷ����������ᣬʹ֮ת��Ϊ����������ˮ�У��ܽ�ӷ��������Ҵ�����������������ˮ�е��ܽ�ȣ����÷ֲ��ȡ�ϲ���������������õ�����Ҫ���������������Ƿ�Һ©����
�ʴ�Ϊ���ϲ㣻CH3COOH+CH3CH2OH$?_{��}^{Ũ����}$CH3COOCH2CH3+H2O����Һ©����
��5�����������ͱ���Na2CO3��Һ��Ӧ���۲쵽���������ݲ�����2CH3COOH+Na2CO3-��2CH3COONa+CO2��+H2O��˵����������Ա�̼��ǿ��
�ʴ�Ϊ����������Ա�̼��ǿ��
���� ���⿼�������������Ʊ���Ϊ��Ƶ���㣬ע��ʵ����Һ�����ơ�����̼������Һ�������Լ�������Ӧ�Ļ���������������ѧ���������������������Ӧ����ѧ֪ʶ���ʵ��������������Ѷ��еȣ�

 53������ϵ�д�
53������ϵ�д�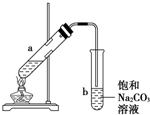 ��ͼΪʵ������ȡ��������������װ��ͼ�����й��ڸ�ʵ��������У�����ȷ���ǣ�������
��ͼΪʵ������ȡ��������������װ��ͼ�����й��ڸ�ʵ��������У�����ȷ���ǣ�������| A�� | ��a�Թ����ȼ����Ҵ���Ȼ���ҡ���Թܱ���������Ũ���ᣬ�ټӱ����� | |
| B�� | �Թ�b�е������¶˹ܿڲ��ܽ���Һ���ԭ���Ƿ�ֹʵ������з����������� | |
| C�� | ʵ��ʱ�����Թ�a��Ŀ���Ǽ�ʱ�����������������ӿ췴Ӧ���� | |
| D�� | �ɽ�����̼������Һ��������������Һ |
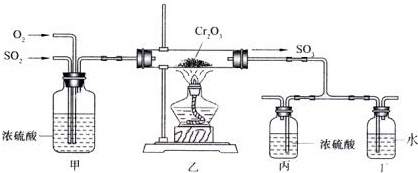
| A�� | ƽ�ⳣ������ | B�� | n��O2������ | ||
| C�� | SO2������������� | D�� | c��SO2������ |
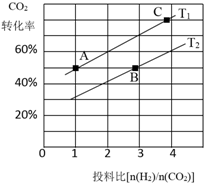 ������̼�IJ����������ҹ���Դ�����һ����Ҫս�Է���
������̼�IJ����������ҹ���Դ�����һ����Ҫս�Է���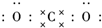 ��������ѧ���������ǹ��ۼ���
��������ѧ���������ǹ��ۼ���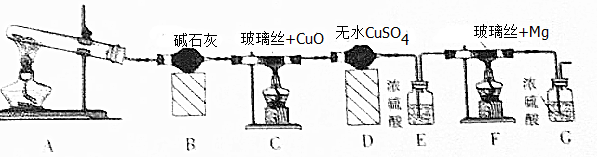
 ��Fe3����I2 B��I2��Cl2��Fe3�� C��Fe3����Cl2��I2 D��Cl2��I2��Fe3��
��Fe3����I2 B��I2��Cl2��Fe3�� C��Fe3����Cl2��I2 D��Cl2��I2��Fe3��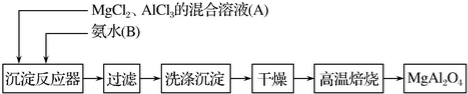
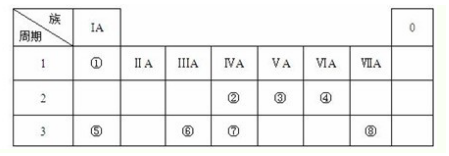
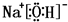 ��
��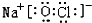 ��
��