��Ŀ����
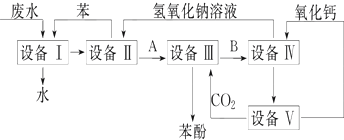
2����ͼ��ʾһЩ�����е�ijЩ�ṹ�����Ƿֱ���NaCl��CsCl���ɱ������ʯ�ṹ��һ��������
��1�����д������ʯ���ǣ�������ĸ����ͬ���٣�����ÿ��̼ԭ����4��̼ԭ����ӽ��Ҿ�����ȣ����ʯ����ԭ�Ӿ��壮
��2�����б�ʾNaCl���Ǣڣ�ÿ��Na+��Χ������ӽ��Ҿ������Na+��12����ÿ��������4��Na+��
��3������CsCl���Ǣۣ����������Ӿ��壬ÿ��Cs+��8��Cl-���ڣ�
��4�������ɱ����Ǣܣ������ڷ��Ӿ��壬ÿ��CO2������12��CO2���ӽ��ڣ��þ�������4��CO2���ӣ�
��5�������������ʵ��۵��ɸߵ��͵�����˳��Ϊ�����������ƣ����ʯ��NaCl��CsCl���ɱ���
���� ���ݲ�ͬ����Ľṹ�ص������ͼ�������������ʣ����ʯ�Ļ�����Ԫ���������壬Ϊ�ռ���״�ṹ��NaCl�����Ǽ�������Ԫ�������ӵ���λ����6���Ȼ�蘆���λ����8��NaCl�����Ǽ�������Ԫ���ɱ�Ҳ��������ṹ������������ÿ��������������붼��һ��CO2���ӣ���Ϊ���������������ɱ��Ƿ��Ӿ��壬�������ʵ��۵��ϵΪԭ�Ӿ��壾���Ӿ��壾���Ӿ��壬���þ�̯�����Խ��о����ļ��㣬�ݴ˷������
��� �⣺���ݲ�ͬ���ʾ���Ľṹ�ص������ͼ�������������ʣ�
��1�����ʯ��ԭ�Ӿ��壬��������ԭ�ӣ��γɵ��ǿռ���״�ṹ����ѡ�٣�����ÿ��̼ԭ����4��̼ԭ����ӽ��Ҿ�����ȣ����ʯ����ԭ�Ӿ��壮
�ʴ�Ϊ���٣�4��ԭ�ӣ�
��2���Ȼ��������Ӿ��壬�乹�������������ӣ�NaCl�����Ǽ�������Ԫ���������Ӽ�ͨ�����Ӽ���ϣ���ѡ�ڣ�ÿ��Na+��Χ������ӽ��Ҿ������Na+��12����ÿ��������12��$\frac{1}{4}$+1=4��Na+��
�ʴ�Ϊ���ڣ�12��4��
��3��CsCl�����������ӹ��ɵģ������Ӻ�����ӵ���λ����8����ѡ�ۣ����������Ӿ��壬ÿ��Cs+��8��Cl-���ڣ��ʴ�Ϊ���ۣ����ӣ�8��
��4���ɱ��Ƿ��Ӿ��壬CO2����λ��������Ķ���������ϣ��Զ����ϵ�CO2����Ϊ�����������������CO2���ӷֲ�����ö���������12����������ϣ�����ͼ��Ϊ�ɱ����壬�þ�������$8��\frac{1}{8}+6��\frac{1}{2}$=4��CO2���ӣ�
�ʴ�Ϊ���ܣ����ӣ�12��4��
��5�������۷е㣺ԭ�Ӿ��壾���Ӿ��壾���Ӿ��壬���Ӿ����۷е������Ӱ뾶�����ȣ����ɳ����ȣ����ʯ��ԭ�Ӿ��塢NaCl��CsCl�����Ӿ��塢�ɱ��Ƿ��Ӿ��壬�����Ӱ뾶С������Ӱ뾶�������⼸�־����۵�ߵ�˳���ǽ��ʯ��NaCl��CsCl���ɱ���
�ʴ�Ϊ�����ʯ��NaCl��CsCl���ɱ���
���� ���⿼���˾������͵��жϣ��ѶȲ����ݲ�ͬ���ʾ���Ľṹ�ص������ͼ������������������ɣ�

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д� �ͱ�Ϊ�����ڡ�ͬ����Ԫ����ɵĵ��ʣ��ҺͶ�����������Ԫ����ɵĻ��������֮������ͼ��ʾ��ת����ϵ�������������ļͱ����������и��������еģ�������
�ͱ�Ϊ�����ڡ�ͬ����Ԫ����ɵĵ��ʣ��ҺͶ�����������Ԫ����ɵĻ��������֮������ͼ��ʾ��ת����ϵ�������������ļͱ����������и��������еģ�������| A�� | H2��Na | B�� | S��O2 | C�� | C��Si | D�� | Cl2��Br2 |
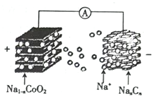 ���������ӵ�ؾ�����Դ�ḻ���ɱ��ͣ�����ת��Ч�ʸߡ����������ƣ�һ����̼�����ϣ�NaxCn���������Ŀɳ�������ӵ�صĹ�ԭ�����£�
���������ӵ�ؾ�����Դ�ḻ���ɱ��ͣ�����ת��Ч�ʸߡ����������ƣ�һ����̼�����ϣ�NaxCn���������Ŀɳ�������ӵ�صĹ�ԭ�����£�NaCoO2+Cn$?_{�ŵ�}^{���}$Na2-xCoO2+NaxCn
���ؽṹ��ͼ��ʾ������˵������ȷ���ǣ�������
| A�� | ���ʱ��Na+�������ƶ� | |
| B�� | �ŵ�ʱ�������ĵ缫��ӦʽΪNaxCn-xe-=xNa++Cn | |
| C�� | ���ʱ������������С | |
| D�� | ���ʱ�������ĵ缫��ӦʽΪNaCoO2-xe-=Na1-xCoO2+xNa+ |
| A�� | X��Y�γɻ�����ʱ��X�Ը��ۣ�Y������ | |
| B�� | �縺��X��Y | |
| C�� | ��ۺ���������ԣ�X��Ӧ����������Y��Ӧ������ | |
| D�� | ʧȥ��һ����������������X��Y |
| A�� | PCl5 ����������ԭ�Ӷ����������8�����ȶ��ṹ�ҹ��õ��ӶԷ���ƫ�� | |
| B�� | ����ԭ�Ӽ��γɹ��ۼ�ʱ���������������Ҽ� | |
| C�� | H3O+��H-O-H�ļ��DZ�H2O��H-O-H�ļ��Ǵ� | |
| D�� | ����ǿ���Ƚϣ�HClO��HClO2 |
| A�� | ʵ�������������Ʊ�����ú���� | |
| B�� | ��﮵�藍����������� | |
| C�� | ��������ˮ��Ӧʱ�ų����� | |
| D�� | ��﮵�藍�������ˮ��ӦԽ��Խ���� |