��Ŀ����
11�����ᾧ�����ɿ���H2C2O4•xH2O��ʾ��Ϊ�˲ⶨxֵ����ȡWg���ᾧ�壬���100.00mlˮ��Һ��Ȼ���������ʵ�飺�ٴӵζ����зų�25.00ml�����ƵIJ�����Һ����ƿ�ڣ���������ϡH2SO4����Ũ��Ϊa mol•L-1��KMnO4��Һ�ζ�����������ΪCO2����������ˮϴ�ӵζ��ܺ���ƿ���۵ζ��ܼ�©�����ô�װҺ��ϴ�ζ��ܣ�����ζ����м����װҺ��������ʼ���������߹������ݣ��Իش���1������ʵ�鲽�����ȷ�����ǣ��ñ�������ۢڢܢݢ٣�
��2���жϵζ��յ�ķ����ǵ��������һ�θ��������Һʱ����Һ����ɫ��Ϊ��ɫ���ұ���30s����ɫ��
��3�����ڽӽ��ζ��յ�ʱ������������ˮ����ƿ�ڱڳ�ϴһ�£��ټ����ζ����յ㣬������õ�xֵ����Ӱ�죨ƫ��ƫС����Ӱ�죩��
��4���ڵζ�����������amol•L-1��KMnO4��ҺVml���ɴ˿ɼ���x��ֵ��$\frac{200w-45aV}{9aV}$��
��5������˫ָʾ���ζ���NaHCO3��Na2CO3��NaOH���������е�һ�ֻ�����������ɵĻ�����и��ɷֵ��������������������ǣ����������Һ�м����̪���ñ�����ζ�����NaOH��Na2CO3��ת��ΪNaCl��NaHCO3ʱ����̪�ɺ�ɫ��Ϊ��ɫ������V1ml���Ȼ��μӼ��ȣ������ñ�����ζ�����NaHCO3ת��ΪNaClʱ����Һ�ɻ�ɫ��Ϊ��ɫ������V2ml���ᣮ����ȡ1.500g �����ʵ����������ʲ������ᷴӦ������100.0mlˮ��Һ��ȡ��20.00ml��Һ����0.1000mol/L�ı�����ζ������V1=35.00ml��V2=5ml���������ijɷּ���������NaOH��69.4%��Na2CO3��30.6%��
���� ��1������������ԭ�ζ��IJ����͵ζ��ܵ�ʹ�ù淶��֪���ζ�ʱ����Ҫ�ζ��ܼ�©��Ȼ��������ˮϴ�ӵζ��ܺ���ƿ���ô�װҺ��ϴ�ζ��ܣ���ζ����м����װҺ��������ʼ���������߹������ݣ��ӵζ����зų�25.00ml�����ƵIJ�����Һ����ƿ�ڣ���������ϡH2SO4����Ũ��Ϊa mol•L-1��KMnO4��Һ�ζ�����������ΪCO2���ݴ˴��⣻
��2����KMnO4��Һ�ζ�������Һ���ζ��յ�ʱ��Һ����ɫ��Ϊ��ɫ����ȥ��
��3���ڽӽ��ζ��յ�ʱ������������ˮ����ƿ�ڱڳ�ϴһ�£���ʵ��û��Ӱ�죻
��4�����ݵ��ӵ�ʧ�غ��й�ϵʽ2KMnO4 ��5H2C2O4���ɼ���ò�������ʵ���������ȷ�����������������Ʒ��������ȥ�����������Ϊ�ᾧˮ���������ݴ˿ɼ����xֵ��
��5�����������֪���Է�̪��ָʾ����ָʾ��ӦΪNaOH+HCl�TNaCl+H2O��Na2CO3+HCl�TNaCl+NaHCO3��������ָʾ����ָʾ��ӦΪ��NaHCO3+HCl�TNaCl+CO2��+H2O������V1��V2��������Ʒ��һ�������������ƣ�����̼���������������Ʒ�Ӧ��������һ����Ϊ̼���ƣ��ٸ��ݷ�Ӧ��Ҫ���������������и��ɷֵ�����������
��� �⣺��1������������ԭ�ζ��IJ����͵ζ��ܵ�ʹ�ù淶��֪���ζ�ʱ����Ҫ�ζ��ܼ�©��Ȼ��������ˮϴ�ӵζ��ܺ���ƿ���ô�װҺ��ϴ�ζ��ܣ���ζ����м����װҺ��������ʼ���������߹������ݣ��ӵζ����зų�25.00ml�����ƵIJ�����Һ����ƿ�ڣ���������ϡH2SO4����Ũ��Ϊa mol•L-1��KMnO4��Һ�ζ�����������ΪCO2������ʵ����Ϊ�ۢڢܢݢ٣�
�ʴ�Ϊ���ۢڢܢݢ٣�
��2����KMnO4��Һ�ζ�������Һ���ζ��յ�ʱ��Һ����ɫ��Ϊ��ɫ����ȥ�������жϵζ��յ�ķ����ǵ��������һ�θ��������Һʱ����Һ����ɫ��Ϊ��ɫ���ұ���30s����ɫ��
�ʴ�Ϊ�����������һ�θ��������Һʱ����Һ����ɫ��Ϊ��ɫ���ұ���30s����ɫ��
��3���ڽӽ��ζ��յ�ʱ������������ˮ����ƿ�ڱڳ�ϴһ�£���ʵ��û��Ӱ�죬
�ʴ�Ϊ����Ӱ�죻
��4��amol•L-1��KMnO4��ҺVml�к��и�����ص����ʵ�ΪaV��10-3 mol�����ݵ��ӵ�ʧ�غ��й�ϵʽ2KMnO4 ��5H2C2O4����֪��������ʵ���Ϊ2.5aV��10-3 mol������ȷ�����������Ϊ255aV��10-3 g�����Խᾧˮ������ΪWg-255aV��10-3 g��������$\frac{18x}{90+18x}=\frac{Wg-255aV��10{\;}^{-3}g}{Wg}$����x=$\frac{200w-45aV}{9aV}$��
�ʴ�Ϊ��$\frac{200w-45aV}{9aV}$��
��5�����������֪���Է�̪��ָʾ����ָʾ��ӦΪNaOH+HCl�TNaCl+H2O��Na2CO3+HCl�TNaCl+NaHCO3��������ָʾ����ָʾ��ӦΪ��NaHCO3+HCl�TNaCl+CO2��+H2O������V1��V2��������Ʒ��һ�������������ƣ�����̼���������������Ʒ�Ӧ��������һ����Ϊ̼���ƣ��������ΪNaOH��Na2CO3����һ�������ķ�ӦΪNaOH+HCl�TNaCl+H2O��Na2CO3+HCl�TNaCl+NaHCO3���ڶ��������ķ�ӦΪNaHCO3+HCl�TNaCl+CO2��+H2O��̼���Ƶ�һ�����ڶ��������ᷴӦ��Ҫ�����ȣ����һ�������������������Ʒ�Ӧ��n��NaOH��=0.1000mol/L����0.035-0.005��L=0.003mol����̼���Ʒ�Ӧ����������Ϊ5mL����n��Na2CO3��=0.1000mol/L��0.005L=0.0005mol��
����NaOH����������Ϊ$\frac{0.003��40}{0.003��40+0.0005��106}$��100%=69.4%��Na2CO3����������Ϊ1-69.4%=30.6%��
�ʴ�Ϊ��NaOH��69.4%��Na2CO3��30.6%��
���� ���⿼�黯ѧ���㼰�к͵ζ���������ԭ�ζ�������������ѡ�ü��ζ�����������Ŀ�Ѷ��еȣ�ע�����յζ������ķ�����ѧ����ݵζ�ԭ�����м������������

| A�� | Ũ�����������ԣ�ϡ����û�������� | |
| B�� | ϡ��Ũ����ʱ����Ũ����������Ͳ������ע��ʢˮ����Ͳ�У������Ͻ��� | |
| C�� | ����Ũ��Խ����������Խǿ | |
| D�� | ���������ᷴӦ��Ũ����Ũ��ϡ����Ӧ�Ļ�ԭ������NO2��NO��H2 |
| A�� | ��һ����KAl��SO4��2��Һ��һ����Ba��OH��2��Һ��ϣ�����������ǡ�����ʱ��Al3++2SO42-+3OH-+2Ba2+�T2BaSO4��+Al��OH��3�� | |
| B�� | ��Ba��OH��2��Һ�еμ�NaHSO4��Һ�������Һǡ��Ϊ���ԣ�Ba2++OH-+H++SO42-�TBaSO4��+H2O | |
| C�� | FeCl3��Һ��Cu�ķ�Ӧ��Cu+Fe3+�TCu2++Fe2+ | |
| D�� | ��Al��OH��3�к����θ�Al��OH��3+3H+�TAl3++3H2O |
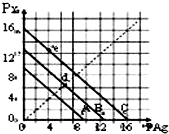 ij�¶�ʱ��±������AgX��X=Cl��Br��I����3���ܽ�ƽ��������ͼ��ʾ��AgCl��AgBr��AgI��Ksp���μ�С����֪pAg=-lgc��Ag+����pX=-lgc��X-��������pX-pAg������ϵ�ɱ�ʾ�� AgX���ܶȻ�����Һ�е�c��Ag+����c��X-�������ϵ������˵��������ǣ�������
ij�¶�ʱ��±������AgX��X=Cl��Br��I����3���ܽ�ƽ��������ͼ��ʾ��AgCl��AgBr��AgI��Ksp���μ�С����֪pAg=-lgc��Ag+����pX=-lgc��X-��������pX-pAg������ϵ�ɱ�ʾ�� AgX���ܶȻ�����Һ�е�c��Ag+����c��X-�������ϵ������˵��������ǣ�������| A�� | e���ʾ�ɹ�����KI��AgNO3��Ӧ����AgI���� | |
| B�� | A����AgCl���ܽ�ƽ�����ߣ�C����AgI���ܽ�ƽ������ | |
| C�� | d������ˮ�м���������AgBr�γɵı�����Һ | |
| D�� | ����㣨8��4���γɵ���Һ��AgCl�IJ�������Һ |
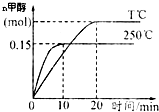 ��1molCO��2molH2����һ�ݻ�Ϊ1L���ܱ������У��ֱ���250�桢T�淢����Ӧ��CO��g��+2H2��g��?CH3OH��g����H=akJ/mol������ͼʾ�жϣ����н�����ȷ���ǣ�������
��1molCO��2molH2����һ�ݻ�Ϊ1L���ܱ������У��ֱ���250�桢T�淢����Ӧ��CO��g��+2H2��g��?CH3OH��g����H=akJ/mol������ͼʾ�жϣ����н�����ȷ���ǣ�������| A�� | a��0��T��250 | |
| B�� | 250��ʱ��0��10min�ڣ�v��H2��=0.015mol/��L•min�� | |
| C�� | CO��ƽ��ת������250��ʱ��T��� | |
| D�� | 250��ʱ����ʼʱ�������иij�1molCH3OH���壬���´�ƽ���CH3OH�����ʵ�������0.15mol |

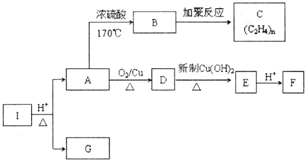 ������I�ķ���ʽΪC6H10O4��75%��A��Һ������ҽ����������I��صķ�Ӧ��ͼ������������Ϣ�ش��������⣮
������I�ķ���ʽΪC6H10O4��75%��A��Һ������ҽ����������I��صķ�Ӧ��ͼ������������Ϣ�ش��������⣮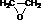 ��
��