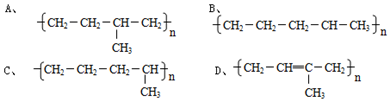��Ŀ����
19�����ʼ��仯�����й㷺Ӧ�ã���1�������������Ϊ�����������֮����ȥ����ˮ���ӵIJ�����������ƣ��׳ơ��������ơ����dz��õ�ˮ���������仯ѧʽΪNa5P3O10��
��2���������ƣ�NaH2PO2�������ڻ�ѧ������
��NaH2PO2��PԪ�صĻ��ϼ�Ϊ+1��
�ڻ�ѧ��������Һ�к���Ni2+��H2PO2-�������������·���������Ӧ������ƽ��
2Ni2++1H2PO2-+1H2O�T2Ni++1H2PO3-+2H+
��3��������뽹̿��ʯӢɰ��ϣ��ڵ�¯�м��ȵ�1 500�����ɰ��ף���ӦΪ
2Ca3��PO4��2+6SiO2$\frac{\underline{\;\;��\;\;}}{\;}$6CaSiO3+P4O10
10C+P4O10$\frac{\underline{\;\;��\;\;}}{\;}$P4+10CO
����Ӧ����31g P4����Ӧ������ת�Ƶĵ�����Ϊ5NA����NA��ʾ�����ӵ�������ֵ����
���� ��1�������������Ϊ�����������֮����ȥ����ˮ���ӵIJ����������Ӽ������ǻ���ȥӦ��ˮ���ӣ����������ƿ��Կ�����������������ǻ�����ԭ�ӻ��������ӣ�
��2���ٸ��ݻ��������ܻ��ϼ�Ϊ0�����PԪ�صĻ��ϼۣ�
�ڲ�ȱ���ӷ���ʽ����ƽ��ע���ʧ�����غ㣬��ƽ��һ��Ҫ������Ƿ��غ㣻
��3������P��+5�۱��0�ۣ�ÿ����1 mol P4ʱ��ת�Ƶ���20 mol�������㣮
��� �⣺��1��������Ӽ�ͨ���ǻ���ˮ�γ��������ᣬ������������ĽṹʽΪ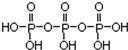 �������������ǻ�����ԭ�ӱ�������ȡ���������������ƣ��׳ơ����ơ������������������ƵĻ�ѧʽΪNa5P3O10���ʴ�Ϊ��Na5P3O10��
�������������ǻ�����ԭ�ӱ�������ȡ���������������ƣ��׳ơ����ơ������������������ƵĻ�ѧʽΪNa5P3O10���ʴ�Ϊ��Na5P3O10��
��2����NaH2PO2�У��ܻ��ϼ�Ϊ0��������Ԫ��Ϊ+1�ۣ���Ԫ��Ϊ-2�ۣ���PԪ�صĻ��ϼ�Ϊ��+1�ۣ��ʴ�Ϊ��+1��
�ڸ��ݵ�ʧ�����غ㣺��Ԫ�صĻ��ϼ۽�����2�ۣ���Ԫ�صĻ��ϼ����ߵ�2�ۣ����Ը���ԭ���غ��ϵ���غ�ɵ���ƽ�ķ���ʽΪ��2Ni2++H2PO2-+H2O=2Ni++H2PO3-+2H+��
�ʴ�Ϊ��2��1��1��H2O��2��1��2H+��
��3��P��+5�۱��0�ۣ�ÿ����1 mol P4ʱ��ת�Ƶ���20 mol��31 g P4�����ʵ���Ϊ0.25 mol�����Է�Ӧת�Ƶĵ�����Ϊ5NA���ʴ�Ϊ��5NA��
���� ���⿼�������ʻ�ѧʽ����д��������ԭ��Ӧ����ʽ����ƽ����ȷ����������ӽṹ�����ʣ���Ϥ������ԭ��Ӧ��ʧ���ӡ�ԭ�Ӹ����غ��ǽ���ؼ�����Ŀ�ѶȲ���

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д� ������أ�KMnO4����Ũ�������Ⱦ��ܷ�����Ӧ����Cl2��������ͼ��ʾ��ʵ��װ���Ʊ������������Cl2�������д����װ���ǣ�������
������أ�KMnO4����Ũ�������Ⱦ��ܷ�����Ӧ����Cl2��������ͼ��ʾ��ʵ��װ���Ʊ������������Cl2�������д����װ���ǣ�������| A�� | �٢� | B�� | �� | C�� | �ڢ� | D�� | �ڢۢ� |
 ����x��y����ԭ��3p�ܼ��ϵĵ������ֱ�Ϊ��������
����x��y����ԭ��3p�ܼ��ϵĵ������ֱ�Ϊ��������| A�� | 18��6��4 | B�� | 22��8��6 | C�� | 26��14��6 | D�� | 15��3��1 |
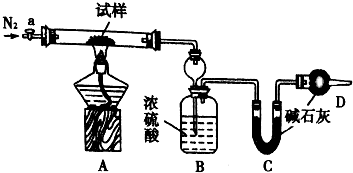 С�մ�NaHCO3�������к���̼���ƾ��壨Na2CO3•l0H2O��Ϊ�ⶨ������С�մ����������w��NaHCO3����ʵ��С��ͬѧ�������ͼװ�ý���ʵ�飮
С�մ�NaHCO3�������к���̼���ƾ��壨Na2CO3•l0H2O��Ϊ�ⶨ������С�մ����������w��NaHCO3����ʵ��С��ͬѧ�������ͼװ�ý���ʵ�飮ʵ����̣�
I����ͼ��װ���������װ�õ������ԣ�
II����m1 g��������Ӳ�ʲ������У�װ��B��C��D��ҩƷ��ͼ����֪��ҩƷ��װ��B������Ϊm2g��װ��C������Ϊm3 g��
�رջ���a����ȼ�ƾ��Ƽ���������ֱ��Bװ����������ð������a��װ����ͨ��N2��һ��ʱ������ƾ��ƣ��رջ���a�������Ƶ�װ��B������Ϊm4g��װ��C������Ϊm5g��
��ش��������⣨װ����ԭ�п�����ʵ���Ӱ����Բ��ƣ���
��1��װ��A�з�����Ӧ�Ļ�ѧ����ʽΪ��2NaHCO3$\frac{\underline{\;\;��\;\;}}{\;}$Na2CO3+CO2��+H2O��Na2CO3•10H2O$\frac{\underline{\;\;��\;\;}}{\;}$Na2CO3+10H2O��
��2��װ��B������Ϊ���շ�Ӧ���ɵ�ˮ������װ��C������Ϊ���շ�Ӧ���ɵĶ�����̼��
��3��ʵ�������ͨ��N2��Ŀ��Ϊʹװ���еĶ�����̼��ˮ����ȫ���ų���װ��BCȫ�����գ�
��4�������и������ṩ�������ܼ����NaHCO3����������w��NaHCO3������bce����ѡ����ĸ��
| ��� | a | b | c | d | e |
| ���� | m1��m2��m3 | m2��m3��m4��m5 | m1��m2��m4 | m1��m4��m5 | m1��m3��m5 |
| A�� | �������ռ���Һ�ķ�Ӧ | B�� | Al��ϡ����ķ�Ӧ | ||
| C�� | C+CO2$\frac{\underline{\;����\;}}{\;}$2CO | D�� | �������������������ֽ� |
| A�� | ��������оƬ | B�� | SiO2�ƹ��ά | ||
| C�� | ���ɰ��̼���裩��ɰ�ֵ�ĥ�� | D�� | ˮ�������ϼ� |
 �ס������ص缫���϶���������̼������ش��������⣺
�ס������ص缫���϶���������̼������ش��������⣺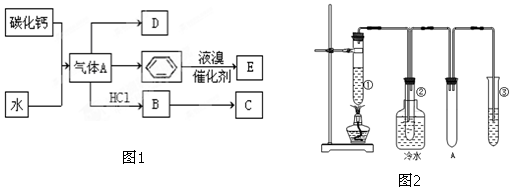
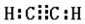 ��
��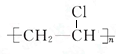 ���䷴Ӧ����Ϊ�Ӿ۷�Ӧ��
���䷴Ӧ����Ϊ�Ӿ۷�Ӧ��