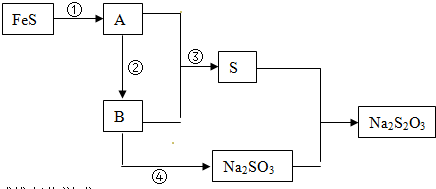
��Ŀ����
12����һ����ĩ���ܺ���K+��Fe3+��Al3+��Cl-��SO42-��CO32-�е������֣��ֽ�������ʵ�飺��ȡ�������壬����ϡ������裬����ȫ���ܽ⣬û������ų���
�������Һ�м���һ����Ba��OH��2��Һ��������ɫ���������˺�����Һ�е����������ữ��AgNO3��Һ���а�ɫ�������ɣ�
��ȡ���е���ɫ��������������ϡ�������ȫ���ܽ⣻
������ȡ�������������������ˮ�������ȫ���ܽ⣬�õ�������Һ��
����ܵ���Һ�м��백ˮʹ��Һ�ʼ��ԣ��г������ɣ����ˣ����õ��ij����м��������NaOH��Һ�����������ܽ⣮
��1����������ʵ�飬�����ĩ��һ�����е�������Fe3+��Al3+��Cl-������ȷ���Ƿ��е����ӿ�ͨ����ɫ��Ӧʵ�飨��ʵ�����ƣ�����һ��ȷ�������ӣ�
��2���������ɳ�����Ӧ���й����ӷ���ʽ��Fe3++3NH3•H2O=Fe��OH��3��+3NH4+��Al3++3NH3•H2O=Al��OH��3��+3NH4+��
��3�����г������ٷ�Ӧ���йػ�ѧ����ʽ��Al��OH��3+NaOH=NaAlO2+2H2O��
���� ��һ����ĩ���ܺ���K+��Fe3+��Al3+��Cl-��SO42-��CO32-�е������֣�������ʵ�飺
��ȡ�������壬����ϡ������裬����ȫ���ܽ⣬û������ų�������Һ��һ������CO32-��
�������Һ�м���һ����Ba��OH��2��Һ��������ɫ����������Һ�к���Fe3+��Al3+��SO42-�����߶��У����˺�����Һ�е���AgNO3��Һ���а�ɫ�������ɣ�����Һ�к���Cl-��
��ȡ���е���ɫ��������������ϡ�������ȫ���ܽ⣬�����ᱵ���������ᣬ����Һ��һ������SO42-��
������ȡ�������������������ˮ�������ȫ���ܽ⣬�õ�������Һ��
����ܵ���Һ�м��백ˮʹ��Һ�ʼ��ԣ��г������ɣ����ˣ����õ��ij����м��������NaOH��Һ���������٣���������������ǿ������������ܣ���˵����Һ�к���Fe3+��Al3+��
ͨ�����Ϸ���֪������ȷ����Һ���Ƿ���K+���Դ������
��� �⣺��һ����ĩ���ܺ���K+��Fe3+��Al3+��Cl-��SO42-��CO32-�е������֣�������ʵ�飺
��ȡ�������壬����ϡ������裬����ȫ���ܽ⣬û������ų�������Һ��һ������CO32-��
�������Һ�м���һ����Ba��OH��2��Һ��������ɫ����������Һ�к���Fe3+��Al3+��SO42-�����߶��У����˺�����Һ�е���AgNO3��Һ���а�ɫ�������ɣ�����Һ�к���Cl-��
��ȡ���е���ɫ��������������ϡ�������ȫ���ܽ⣬�����ᱵ���������ᣬ����Һ��һ������SO42-��
������ȡ�������������������ˮ�������ȫ���ܽ⣬�õ�������Һ��
����ܵ���Һ�м��백ˮʹ��Һ�ʼ��ԣ��г������ɣ����ˣ����õ��ij����м��������NaOH��Һ���������٣���������������ǿ������������ܣ���˵����Һ�к���Fe3+��Al3+��
ͨ�����Ϸ���֪������ȷ����Һ���Ƿ���K+��
��1������������֪������Һ��һ��������������CO32-��SO42-��һ�����е�������Fe3+��Al3+��Cl-������ȷ���Ƿ��е�������K+��Ҫ����KԪ����Ҫͨ����ɫ��Ӧ���ʴ�Ϊ��Fe3+��Al3+��Cl-����ɫ��Ӧ��
��2���������ɳ�����Ӧ���й����ӷ���ʽΪFe3++3NH3•H2O=Fe��OH��3��+3NH4+��Al3++3NH3•H2O=Al��OH��3��+3NH4+��
�ʴ�Ϊ��Fe3++3NH3•H2O=Fe��OH��3��+3NH4+��Al3++3NH3•H2O=Al��OH��3��+3NH4+��
��3�����������������������Ʒ�Ӧ���ɿ�����ƫ�����ƶ����³������٣���ѧ����ʽΪAl��OH��3+NaOH=NaAlO2+2H2O���ʴ�Ϊ��Al��OH��3+NaOH=NaAlO2+2H2O��
���� ���⿼�������ӷ�Ӧ�������ӵ��ƶϣ�Ϊ��Ƶ���㣬�������ɵij�������������ɫ�����ӹ���ͷ�Ӧȥȷ�����ڵ�����Ϊ���Ĺؼ���ע�����ճ������ӵ����ʼ����鷽������Ŀ�Ѷ��еȣ�

| A�� | 20.7% | B�� | 22% | C�� | 1.3% | D�� | 7.3% |
| A�� | 18gNH4+�������ӵ���ĿΪ10NA | |
| B�� | 1molNa2O2������CO2��Ӧʱ��ת�Ƶ��ӵ���ĿΪ2NA | |
| C�� | ��״���£������Ϊ22.4L��O2��HCl��H2O���еķ�����Ŀ��ΪNA | |
| D�� | ����Ϊ16g��O2��O3�Ļ��������������ԭ�ӵ���ĿΪNA |
| A�� | ��Һ�ʻ���ɫ���Ҿ��д̼�����ζ��˵��Cl2�Ĵ��� | |
| B�� | ������ɫ��������ɫ������ɫ��˵����Һ����Cl2���� | |
| C�� | ����NaHCO3��Һ������ɫ���������˵����HClO���� | |
| D�� | �ȼ��������ữ���ټ�AgNO3��Һ������ɫ������˵����Cl-���� |
| A�� | �����˷�Ӧ�Ļ�� | B�� | �����˷�Ӧ������������ | ||
| C�� | �����˷�Ӧ���ʱ� | D�� | �����˷�Ӧ��ƽ�ⳣ�� |
| A�� | �ڵ������Һ��ע��һ�������������ʹ��Һ�ָ���ԭʼ״̬ | |
| B�� | �ڵ������Һ��ͨ������4.48L���Ȼ������岢ע��0.1mol��ˮ����ʹ��Һ�ָ���ԭʼ״̬ | |
| C�� | ������������������ʵ���������������������ʵ�����0.75�� | |
| D�� | ���������������ƽ��Ħ������Ϊ58g/mol |
���ò�˿��ȡ������Һ���ڻ��������գ�����ɫ�ܲ������۲쵽��ɫ����
����ȡԭ��Һ����������������ɫ�������ɣ���������������ɺ���ɫ����ʱ��Һ��ɫ�������������
��ȡ��Ӧ����Һ�ֱ�������֧�Թ��У���һ֧�Թ��м���BaCl2��Һ�а�ɫ�������ɣ��ٵμ�KSCN��Һ���ϲ���Һ��죬�ڶ�֧�Թܼ���CCl4��������ú���Һ�ֲ㣬�²�Ϊ��ɫ��
����˵����ȷ���ǣ�������
| A�� | ԭ��Һ�п϶�����Fe2+��NO3-��SiO32-��I- | |
| B�� | ԭ��Һ�п϶�����K+��Fe3+��Fe2+��NO3-��SO42- | |
| C�� | ���������ɫ������NO���壬��CO2������� | |
| D�� | Ϊȷ���Ƿ���Cl-��ȡԭ��Һ���������������Һ���۲��Ƿ������ɫ���� |
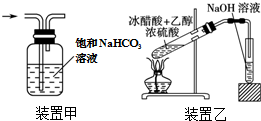
| A�� | ��25.0gCuSO4•5H2O����100mL����ˮ�����1.0mol•L-1����ͭ��Һ | |
| B�� | ��ɫ��Ӧʵ���У���պȡ������Һǰ������ϡ����ϴ����˿������������Ϊ��ɫ | |
| C�� | ��װ�ü׳�ȥCl2�е�HCl���� | |
| D�� | ��װ������ȡ�������� |