��Ŀ����
��̬ũҵ�漰ũ�ҷ��ϵ��ۺ����ã�ij�ַ��Ͼ����͵õ�һ�ֺ����顢������̼�������Ļ�����塣2.016 L����״����������ͨ��ʢ�к���CuO��ĩ��Ӳ�ʲ����ܣ������ķ�ӦΪ��CH4+4CuO![]() CO2+2H2O+4Cu����������ȫ��Ӧ��Ӳ�ʲ����ܵ���������4.8g������Ӧ�����������ͨ�������ij���ʯ��ˮ�У�������գ����ɳ���8.5 g��
CO2+2H2O+4Cu����������ȫ��Ӧ��Ӳ�ʲ����ܵ���������4.8g������Ӧ�����������ͨ�������ij���ʯ��ˮ�У�������գ����ɳ���8.5 g��
��1��ԭ��������м�������ʵ�����_____________��
��2��ԭ��������е������������Ϊ���٣���д��������̣�
��1��0.075 mol (2��5.56%
��������1��Ӳ�ʲ�������������4.8 g������CuOת��ΪCuʱ��ʧȥ��OԪ�ص����������й�ϵʽ��CH4�D4CuO�D4Cu�D4O
n(O)=4.8 g��16 g?mol-1=0.3 mol����n(CH4)=0.3 mol��4=0.075 mol
��2���⣺�������⣬n��CH4+CO2��=n����CO2��=n��CaCO3��
��n��N2��=2.016 L��22.4 L?mol-1-8.5 g��100 g?mol-1=0.005 mol
����ԭ���������N2���������Ϊ(0.005 mol��0.09 mol)��100%=5.56%��

��ϰ��ϵ�д�
�����Ŀ
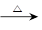 CO2����2H2O����4Cu����������ȫ��Ӧ��Ӳ�ʲ����ܵ�����������4.8g������Ӧ������ͨ������ij���Ca(OH)2��Һ��������գ����ɳ���10g��
CO2����2H2O����4Cu����������ȫ��Ӧ��Ӳ�ʲ����ܵ�����������4.8g������Ӧ������ͨ������ij���Ca(OH)2��Һ��������գ����ɳ���10g�� �ۺ����á�ij�ַ��Ͼ����͵õ�һ�ֺ��м��顢������̼�������Ļ����
�ۺ����á�ij�ַ��Ͼ����͵õ�һ�ֺ��м��顢������̼�������Ļ���� ��8.96L����״������
��8.96L����״������ ������ͨ��ʢ�к�ɫCuO��ĩ��Ӳ�ʲ����ܣ������ķ�ӦΪ��CH4��4CuO
������ͨ��ʢ�к�ɫCuO��ĩ��Ӳ�ʲ����ܣ������ķ�ӦΪ��CH4��4CuO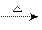 CO2����2H2O����4Cu����������ȫ��Ӧ��Ӳ�ʲ����ܵ�����������4.8g������Ӧ������ͨ��2L 0.1mol/L�ij���Ca(OH)2��Һ��������գ����ɳ���10g��
CO2����2H2O����4Cu����������ȫ��Ӧ��Ӳ�ʲ����ܵ�����������4.8g������Ӧ������ͨ��2L 0.1mol/L�ij���Ca(OH)2��Һ��������գ����ɳ���10g��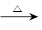 CO2����2H2O����4Cu����������ȫ��Ӧ��Ӳ�ʲ����ܵ�����������4.8g������Ӧ������ͨ������ij���Ca(OH)2��Һ��������գ����ɳ���10g��
CO2����2H2O����4Cu����������ȫ��Ӧ��Ӳ�ʲ����ܵ�����������4.8g������Ӧ������ͨ������ij���Ca(OH)2��Һ��������գ����ɳ���10g��