��Ŀ����
���ԭ���ڻ�ѧ��ҵ���й㷺Ӧ�á���ͼ��ʾһ�����أ�װ�е��Һa��X��Y������缫�壬ͨ��������ֱ����Դ�����������ʷ����ĵ缫��Ӧ����д������ش��������⣺
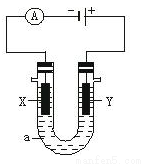
��1����X��Y���Ƕ��Ե缫��a�DZ���NaCl��Һ��ʵ�鿪ʼʱ��ͬʱ�����߸����뼸�η�̪��Һ����
��д����ⱥ��NaCl��Һ�ܵ����ӷ���ʽ ��
����X�������۲쵽��������_______________________������ٵ缫��Ӧ����ķ�����________________��
��2����Ҫ�õ�ⷽ��������ͭ������п��������Ͳ��������ʣ������Һaѡ��CuSO4��Һ����
��X�缫�����ķ�Ӧ����Ϊ ����Y�缫�IJ�����_____________����Ҫ�缫��ӦʽΪ_______________��CuSO4��Һ��Ũ�� ���� �����䡱���������ӡ������м��١���
��ϰ��ϵ�д�
�����Ŀ
18�������ĺ����̲��ŷḻ�Ļ�ѧ��Դ�����м�ʮ��Ԫ�أ��������ں�ˮ����Ԫ�ص��ǣ�������
| A�� | �� | B�� | �� | C�� | �� | D�� | �� |
5����ָ����Һ�У����и����е����Ӳ��ܴ���������ǣ�������
| A�� | ��ˮ�������c��H+��=1��10-12mol/L����Һ��Al3+��CH3COO-��NH4+��HCO3- | |
| B�� | ��KSCN��Һ������Һ��Na+��SO42-��H2O2��Cl- | |
| C�� | ʹ��̪������Һ��Ag��NH3��2+��CO32-��K+��S2- | |
| D�� | $\frac{c��{H}^{+}��}{c��O{H}^{-}��}$=10-12����Һ��K+��AlO2-��SiO32-��Na+ |


 ��
��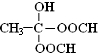 ������֪��
������֪�� ��
��