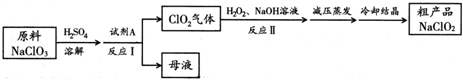
��Ŀ����
8��ʵ����������װ�òⶨFeO��Fe2O3����������Fe2O3��������Dװ�õ�Ӳ��˫ͨ�������еĹ���������FeO��Fe2O3�Ļ���
��1��װ��A�з�����Ӧ�����ӷ���ʽZn+2H+=Zn2++H2��
��2��װ��A�����ķ�Ӧ��ʱҪ�����м�������CuSO4��Һ����Ŀ���Ǽӿ������������ʣ���ԭ����Zn����CuSO4��Ӧ����Cu������Zn���棬Zn����������Cu����������ϡ���ᣨ�������Һ����ɺܶ�Сԭ��أ�����ԭ��ط�Ӧ���ӿ��˷�Ӧ���ʣ�
��3��Ϊ�˰�ȫ���ڵ�ȼD���ľƾ���֮ǰ����b���ڴ������ռ������鴿��
��4��װ��B�������dz�ȥ�����е��Ȼ��⣻װ��C��װ��Һ����Ũ���ᣬ����������Ǹ���������
��5������������ã����ҽ����˱�Ҫ�İ�ȫ������ȼD���ľƾ��ƣ���Ӳ��˫ͨ�������з�����Ӧ�Ļ�ѧ����ʽ��3H2+Fe2O3 $\frac{\underline{\;����\;}}{\;}$2Fe+3H2O��H2+FeO $\frac{\underline{\;����\;}}{\;}$Fe+H2O��
��6����FeO��Fe2O3�������������Ϊ23.2g����Ӧ��ȫ��U�ܵ���������7.2g����������Fe2O3������Ϊ16g��
��7��U��E�ұ������Ӹ����F��Ŀ���Ƿ�ֹ�����е�ˮ�������������E�У��������F�����Fe2O3��������ƫ���ƫ��ƫС������Ӱ�족����
��8������Ӧ��õ��IJ��������л�������FeO����õ�Fe2O3������ƫС���ƫ��ƫС������Ӱ�족����
���� װ��A���Ʊ������ķ���װ�ã�ͨ��װ��B�е�ˮ��ȥ�����е��Ȼ��⣬ͨ��װ��C�е�Ũ�������������ͨ��װ��D��ԭFeO��Fe2O3�Ļ������ɵ�ˮ��������װ��E���գ����������仯�õ�����ˮ������������������������װ��F�Ƿ�ֹ�����е�ˮ��������װ��EӰ��ⶨ�����ȷ�ԣ�
��1������װ��������ѹǿ�仯��Һ��仯�����ж�װ�������ԣ�
��2��Zn����CuSO4��Ӧ����Cu������Zn���棬Zn����������Cu����������ϡ���ᣨ�������Һ����ɺܶ�Сԭ��أ�
��3�������ǿ�ȼ��������Ȼ��ȼǰ��Ҫ�����鴿��
��4��A�в�����H2�л���HCl��ˮ����������ͨ��ϴ����һ��ȥ����ֹ���ź���ʵ�飻
��5��Dװ�÷����ķ�Ӧ��������ԭ�����������������ķ�Ӧ������������ˮ��
��6��������������������������������U�ܵ���������7.2gΪˮ��������ʽ���㣻
��7���������F�������е�ˮ������E��������������ˮ������������Ԫ�����ʵ���ƫ��Fe2O3������Ҳ��ƫ��
��8������Ӧ��õ��IJ��������л�������FeO��E��������������ˮ��������С����Ԫ�����ʵ���ƫС��
��� �⣺��1��A��п��ϡ���ᷴӦ�����Ȼ�п����������Ӧ�����ӷ���ʽΪ��Zn+2H+=Zn2++H2����
�ʴ�Ϊ��Zn+2H+=Zn2++H2������
��2��װ��A�����ķ�Ӧ��ʱҪ�����м�������CuSO4��Һ��п������ͭ��Һ��Ӧ����ͭ������п���棬��ϡ�������γ�ԭ��ط�Ӧ�ӿ췴Ӧ���ʣ�
�ʴ�Ϊ���ӿ������������ʣ�Zn����CuSO4��Ӧ����Cu������Zn���棬Zn����������Cu����������ϡ���ᣨ�������Һ����ɺܶ�Сԭ��أ�����ԭ��ط�Ӧ���ӿ��˷�Ӧ���ʣ�
��3�������ǿ�ȼ�����壬װ������Ҫ���������ɽ��м��ȷ�Ӧ��������Ҫ�����������鴿��������п������ȷ�����ը����F���ռ������鴿��
�ʴ�Ϊ���ռ������鴿��
��4������A�в�����H2�л���HCl��ˮ����������ͨ��ϴ����һ��ȥ����ֹ���ź���ʵ�飬����B�������dz�ȥ�����е��Ȼ��⣬C��Ũ���ᣬ���ڸ���������
�ʴ�Ϊ����ȥ�����е��Ȼ��⣻Ũ�������������
��5��װ��D�з������������������������������ķ�Ӧ����Ӧ����ʽΪ��3H2+Fe2O32Fe+3H2O��H2+FeO$\frac{\underline{\;����\;}}{\;}$Fe+H2O��
�ʴ�Ϊ��3H2+Fe2O3$\frac{\underline{\;����\;}}{\;}$2Fe+3H2O��H2+FeO$\frac{\underline{\;����\;}}{\;}$Fe+H2O��
��6��U�ܵ��������ӵ�7.2g��ˮ�����ʵ����� $\frac{7.2g}{18g/mol}$=0.4mol��
��FeO��Fe2O3���ʵ����ֱ�ΪX��Y����X+3Y=0.4
������������ʽ��72X+160Y=23.2
���Y=0.1��X=0.1��
������������������160g/mol��0.1mol=16g��
�ʴ�Ϊ��16��
��7�������F�����÷�������ˮ������ʵ����ţ��������F�������е�ˮ������E��������������ˮ���������������ĺ�������Fe2O3��������ƫ��
�ʴ�Ϊ����ֹ�����е�ˮ�������������E�У�ƫ��
��8������Ӧ��õ��IJ��������л�������FeO��E��������������ˮ��������С����Ԫ�����ʵ���ƫС�����ݣ�6���н�����֪���ʱ����ˮ����Ϊm��
72X+160Y=23.2
X+3Y=$\frac{m}{18}$
Y=$\frac{56m}{18}$-23.2��mԽС��YԽС��
��õ�Fe2O3������ƫС��
�ʴ�Ϊ��ƫС��
���� ���⿼���˲ⶨFeO��Fe2O3����������Fe2O3����������һ���ۺ�ʵ���⣬����Ĺؼ������ո���װ�õ����ã����ܸ�����ص����ݽ��м��㣬�����ѵ��ѧ��˼ά������һ�������������Ѷ��еȣ�

 ����С��ʿ���������ϵ�д�
����С��ʿ���������ϵ�д�| A�� | ͬһ֧�ֱ�ʹ�ò�ͬƷ��īˮʱ���������������� | |
| B�� | ���չ�Ӵ�϶���밵��ʱ���ɹ۲쵽һ������ | |
| C�� | ������������ˮ�γɵ�ij��ɢϵ�в����������缫��ֱͨ����һ��ʱ���һ��������ɫ���� | |
| D�� | ���Ȼ�����Һ�м�������������Һ���������ɫ���� |
| A�� | ʯ���ﺬ��C5��C11������������ͨ��ʯ�͵ķ���õ����� | |
| B�� | ����C18���ϵ����������;������ѻ����Եõ����� | |
| C�� | ʯ�͵ķ�����һ���������̣����ѻ����ѽ����ڻ�ѧ���� | |
| D�� | ʯ���ѽ��ΩһĿ���ǻ����ϩ |
| A�� | HBr��HCl��HF | B�� | K��Mg��Na | ||
| C�� | MgO��NaCl��KBr | D�� | ����衢̼���衢���ʯ |
| A�� | 1mol��Ϊ32g | |
| B�� | Ħ�������ʵ����ĵ�λ | |
| C�� | 44gCO2�����Ϊ22.4L | |
| D�� | 2mol/L��BaCl2��Һ�к�Cl-�ĸ���Ϊ2.408��1024 |
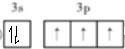 ��������Ӧ�漰�ĵڶ�����Ԫ���е�һ�������ɴ�С��˳��ΪF��O��C����Ԫ�ط��ű�ʾ����
��������Ӧ�漰�ĵڶ�����Ԫ���е�һ�������ɴ�С��˳��ΪF��O��C����Ԫ�ط��ű�ʾ����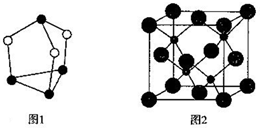
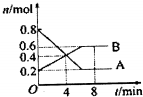 ij�¶�ʱ����0.5L�ܱ�������ijһ��Ӧ��A��B����������ʱ��仯��������ͼ��ʾ������������⣺
ij�¶�ʱ����0.5L�ܱ�������ijһ��Ӧ��A��B����������ʱ��仯��������ͼ��ʾ������������⣺