��Ŀ����
������һ����Ҫ��ҵԭ�ϣ��ڹ�ũҵ�����о�����Ҫ��Ӧ�ã�
��1����֪��N2��g��+O2��g���T2NO��g������H=+180.5kJ•mol-1
4NH3��g��+5O2��g���T4NO��g��+6H2O��g������H=-905kJ•mol-1
2H2��g��+O2��g���T2H2O��g������H=-483.6kJ•mol-1
��N2��g��+3H2��g���T2NH3��g������H=______
��2����ҵ�ϳɰ����ķ�ӦΪN2��g��+3H2��g�� 2NH3��g������һ���¶��£���һ������N2��H2ͨ�뵽��ʼ���Ϊ1L���ܱ������дﵽƽ������ı�������������ʹƽ��������Ӧ�����ƶ���ƽ�ⳣ���������______��������ţ�
2NH3��g������һ���¶��£���һ������N2��H2ͨ�뵽��ʼ���Ϊ1L���ܱ������дﵽƽ������ı�������������ʹƽ��������Ӧ�����ƶ���ƽ�ⳣ���������______��������ţ�
������ѹǿ ������Ӧ���Ũ�� ��ʹ�ô��� �ܽ����¶�
��3������ͬ�����pH֮��Ϊ14�İ�ˮ�������Ϻ���Һ������Ũ���ɴ�С��˳��Ϊ______��
��4�������Ǻϳɰ���ԭ��֮һ����ͼ����SO2��I2��H2OΪԭ�ϣ����ú���ʹˮ�ֽ���������һ�����̡�
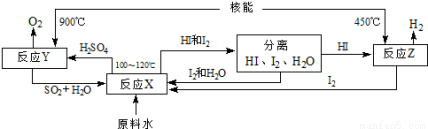
��ӦX�Ļ�ѧ����ʽΪSO2+I2+2H2O=2HI+H2SO4��������ѭ��ʹ�õ�ԭ�ϳ���SO2�⣬���� ���������Ͻ����������У�1molԭ��ˮ�Ƶ� molH2��
�±�������ͬ�¶����������һЩ���ݣ������ж���ȷ���ǣ� ��
�� | HX | HY | HZ | ||
Ũ�ȣ�mol/L�� | 0.12 | 0.2 | 0.9 | 1 | 1 |
����� | 0.25 | 0.2 | 0.1 | 0.3 | 0.5 |
���볣�� | K1 | K2 | K3 | K4 | K5 |
A������ͬ�¶ȣ���HX�����ݿ���˵�������������Һ��Ũ��Խ�ͣ������Խ����K1>K2>K3=0.01
B������ʱ������NaZ��Һ�м�ˮ����c(Z?)/[c(HZ)?c(OH?)]�ı�ֵ��С�������������ᣬ���ֵ���
C�������ʵ�����NaX��NaY��NaZ�Ļ�ϣ�c(X?)+c(Y?)?2c(Z?)=2c(HZ)?c(HX)?c(HY),��c(Z?)<c(Y?)<c(X?)
D������ͬ�¶��£�K5>K4>K3




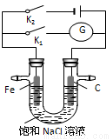
 2C(g)+2D(g)�ڲ�ͬ����²�÷�Ӧ���ʣ����з�Ӧ������
2C(g)+2D(g)�ڲ�ͬ����²�÷�Ӧ���ʣ����з�Ӧ������