��Ŀ����
���г��������µļס��ҡ���������Һ����Ϊ0��1mol��L-1��NaOH��Һ����Ϊ 0��1mol��L-1��HCl��Һ����Ϊ0��1mol��L-1��CH3COOH��Һ���Իش���������
��1������Һ�У���ˮ�������c(H+)��____________ mol��L-1��
��2������Һ�д��ڵĵ���ƽ��Ϊ________________________________���õ���ƽ�ⷽ��ʽ��ʾ����
��3���ס��ҡ���������Һ����ˮ�������c(OH-)�Ĵ�С��ϵΪ____________��
��4��ijͬѧ�ü���Һ�ֱ�ζ�20��00mL����Һ��20��00mL����Һ���õ���ͼ��ʾ�����ζ����ߣ�������й�����
��1������Һ�У���ˮ�������c(H+)��____________ mol��L-1��
��2������Һ�д��ڵĵ���ƽ��Ϊ________________________________���õ���ƽ�ⷽ��ʽ��ʾ����
��3���ס��ҡ���������Һ����ˮ�������c(OH-)�Ĵ�С��ϵΪ____________��
��4��ijͬѧ�ü���Һ�ֱ�ζ�20��00mL����Һ��20��00mL����Һ���õ���ͼ��ʾ�����ζ����ߣ�������й�����

�ټ���Һ�ζ�����Һ��������_____________���ͼ1����ͼ2������
��a��____________mL��
��a��____________mL��
��1��10-13
��2��CH3COOH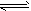 CH3COO-+H+�� H2O
CH3COO-+H+�� H2O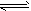 OH-+H+
OH-+H+
��3��������=��
��4����ͼ2����20.00
��2��CH3COOH
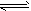 CH3COO-+H+�� H2O
CH3COO-+H+�� H2O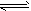 OH-+H+
OH-+H+��3��������=��
��4����ͼ2����20.00

��ϰ��ϵ�д�
�����Ŀ

 l��Һ����Ϊ0.1mol��L-1��CH3COOH��Һ���Իش��������⣺
l��Һ����Ϊ0.1mol��L-1��CH3COOH��Һ���Իش��������⣺ ����й����⣺
����й����⣺

