��Ŀ����
���ܱ������н��е����·�Ӧ��2SO2(g)+O2(g)![]() 2SO3(g)
2SO3(g)
SO2����ʼŨ����0.4 mol��L-1,O2����ʼŨ����1 mol��L-1����SO2��ת����Ϊ80%ʱ����Ӧ�ﵽƽ��״̬��
(1)��Ӧ��ƽ�ⳣ����
(2)����ƽ��ʱ��Ӧ������ѹǿ����1����ƽ�⽫����ƶ���
(3)��ƽ��ʱ��Ӧ������ѹǿ��С1����ƽ�⽫����ƶ���
(4)ƽ��ʱ����������䣬��ƽ���������г���ϡ������Ar,ʹ��ϵ��ѹ��Ϊԭ����3����ƽ���ֽ�����ƶ���
������ 2SO2(g) + O2(g)![]() 2SO3(g)
2SO3(g)
2 1 2
��ʼʱ 0.4 1 0
ƽ��ʱ0.4��(1-80%) 1-![]() ��0.4��80% 0.4��80%
��0.4��80% 0.4��80%
=0.08 =0.84 =0.32
(1)ƽ�ⳣ��K=![]() =
=![]() ��19��
��19��
(2)ѹǿ����1��������ֵ�Ũ������1����
Qc=![]() =
=![]() ��
��![]() ��
��
��Qc��K������ƽ��������Ӧ�����ƶ���
(3)ѹǿ��С1��������ֵ�Ũ��Ҳ��С1����
Qc=![]() =
=![]() ��
��![]() ��
��
��Qc��K������ƽ�����淴Ӧ�����ƶ���
(4)����������䣬����ϡ������Ar��������ѹ�ı䣬����Ӧ��������ֵ�Ũ�ȱ��ֲ��䡣
Qc=![]() =
=![]() =K������ƽ�ⲻ�����ƶ���
=K������ƽ�ⲻ�����ƶ���
�𰸣�(1)K��19��
(2)������Ӧ�����ƶ���
(3)���淴Ӧ�����ƶ���
(4)���ƶ���

 ��֪ij���淴Ӧ��mA��g��+nB��g��?pC��g����H=Q kJ?mol-1�����ܱ������н��У���ͼ��ʾ�ڲ�ͬʱ��t���¶�T��ѹǿP�뷴Ӧ��B�İٷֺ����Ĺ�ϵ���ߣ������ж���ȷ���ǣ�������
��֪ij���淴Ӧ��mA��g��+nB��g��?pC��g����H=Q kJ?mol-1�����ܱ������н��У���ͼ��ʾ�ڲ�ͬʱ��t���¶�T��ѹǿP�뷴Ӧ��B�İٷֺ����Ĺ�ϵ���ߣ������ж���ȷ���ǣ�������| A��T2��T1?P1��P2 m+n��p??������0 | B��T1��T2?P1��P2 m+n��p??������0 | C��T1��T2?P1��P2 m+n��p??������0 | D��T1��T2?P1��P2 m+n��p??������0 |
 qC��g�����ܱ������н��У���ͼ��ʾ��Ӧ�ڲ�ͬʱ��t���¶�T��ѹǿP�뷴Ӧ��B����������Ĺ�ϵ���ߣ�����ͼ����գ���1������2������3�������������������=������
qC��g�����ܱ������н��У���ͼ��ʾ��Ӧ�ڲ�ͬʱ��t���¶�T��ѹǿP�뷴Ӧ��B����������Ĺ�ϵ���ߣ�����ͼ����գ���1������2������3�������������������=������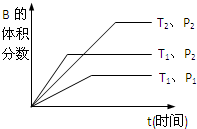
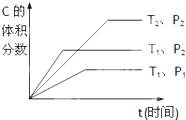 ��֪ij���淴ӦmA��g��+nB��g��?qC��g�����ܱ������н��У���ͼ��ʾ��Ӧ�ڲ�ͬʱ��t���¶�T��ѹǿP�뷴Ӧ��B����������Ĺ�ϵ���ߣ�����ͼ����գ����������������=��������1���¶ȵĹ�ϵ��T1
��֪ij���淴ӦmA��g��+nB��g��?qC��g�����ܱ������н��У���ͼ��ʾ��Ӧ�ڲ�ͬʱ��t���¶�T��ѹǿP�뷴Ӧ��B����������Ĺ�ϵ���ߣ�����ͼ����գ����������������=��������1���¶ȵĹ�ϵ��T1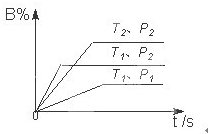 ��֪ij���淴Ӧm A��g��+n B��g��?p C��g����H�����ܱ������н��У���ͼ��ʾ�ڲ�ͬ��Ӧʱ��t ʱ�¶�T��ѹǿP�뷴Ӧ��B�ڻ�������еİٷֺ���B%�Ĺ�ϵ���ߣ������߷����������ж���ȷ���ǣ�������
��֪ij���淴Ӧm A��g��+n B��g��?p C��g����H�����ܱ������н��У���ͼ��ʾ�ڲ�ͬ��Ӧʱ��t ʱ�¶�T��ѹǿP�뷴Ӧ��B�ڻ�������еİٷֺ���B%�Ĺ�ϵ���ߣ������߷����������ж���ȷ���ǣ������� ��֪ij���淴ӦmA��g��+nB��g��?pC��g�������ܱ������н��У���ͼ��ʾ�ڲ�ͬ��Ӧʱ��t���¶�T��ѹǿP�뷴Ӧ��B�ڻ�������еİٷֺ���B%�Ĺ�ϵ���ߣ������߷����������ж���ȷ���ǣ�������
��֪ij���淴ӦmA��g��+nB��g��?pC��g�������ܱ������н��У���ͼ��ʾ�ڲ�ͬ��Ӧʱ��t���¶�T��ѹǿP�뷴Ӧ��B�ڻ�������еİٷֺ���B%�Ĺ�ϵ���ߣ������߷����������ж���ȷ���ǣ�������