��Ŀ����
(16��)��ͭ�����Ҫ�ɷ���CuFeS2(��Ԫ���ԡ�2�ۣ���Ԫ����+2��)��ʵ�������û�ͭ��Ϊԭ����ȡ����ͭ������(Fe2O3)���������£�

��֪��CuFeS2+O2 Cu+FeS+ SO2 ��FeS+2HCl
Cu+FeS+ SO2 ��FeS+2HCl FeCl2+H2S��
FeCl2+H2S��
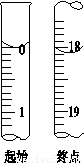
��1����ʵ�����У�Ӧ����ͭ���ĩ����_____(����������)�б��ա�
��2������Ӧ�����в�����SO2��H2Sͨ����ͼ��ʾװ���м������ǵ����ʡ���ʵ��֤��SO2����_________�Ժ�__________�ԡ�

��3����ѡ�����в���װ����ʵ��������MnO2��Ũ����Ϊԭ����ȡ�����������������
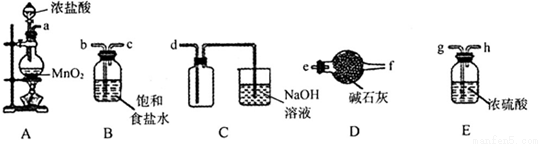
��Բ����ƿ�з�����Ӧ�����ӷ���ʽΪ_________________________________________��
�ڰ��������������ң����ܿ�����˳��Ϊa��________________________________��
��װ�����Ӻú���Ҫ���еIJ�����_________________��
��4��������Fe2O3��Ʒ�к���FeO��ѡ���ṩ���Լ������ʵ����֤�����к���FeO��д���й�ʵ���������������ۡ�________________________________________________________��
�ṩ���Լ���ϡ���ᡢϡ���ᡢKSCN��Һ��KMnO4��Һ��NaOH��Һ����ˮ��
��1��������2�֣�
��2��Ư�ף�2�֣� ������2�֣�
��3�� ��MnO2+4H++2Cl? Mn2++Cl2��+2H2O��3�֣�
Mn2++Cl2��+2H2O��3�֣�
��b��c��g��h��d��2�֣�
�ۼ��װ�õ������� ��2�֣�
��4��ȡ����������Ʒ���Թ��У���ϡ�����ܽ⣬����Һ�еμ�����KMnO4��Һ����KMnO4��Һ�Ϻ�ɫ��ɫ����֤������FeO ��3�֣�
��������
�����������1����ʵ�����й���������������ڽ��У�Ӧ����ͭ���ĩ���������б��ա���2��SO2ͨ��Ʒ����Һ��Ʒ����Һ��ɫ��֤��SO2����Ư���ԣ�SO2ͨ��H2S��Һ������S����ɫ������֤��SO2���������ԡ���3����MnO2��Ũ�����ڼ��������·�Ӧ����Cl2��MnCl2��H2O�����ӷ���ʽΪ��MnO2+4H++2Cl? Mn2++Cl2��+2H2O�������ɵ�Cl2����HCl��H2O������a������b��c��ͨ������ʳ��ˮ��ȥHCl��������g��h��ͨ��Ũ���ᣬ��ȥH2O��������d�������ռ��������������������װ�����Ӻú���Ҫ���еIJ����Ǽ��װ�õ������ԣ���4����֤�����к���FeO�����������������ӵĻ�ԭ�ԣ��������Լ�������ʵ�����Ϊ��ȡ�����������ӹ���ϡ�����ܽ⣬Ȼ������Һ�м�����KMnO4��Һ������Һ��ɫ��ȥ����˵�������к���FeO��
Mn2++Cl2��+2H2O�������ɵ�Cl2����HCl��H2O������a������b��c��ͨ������ʳ��ˮ��ȥHCl��������g��h��ͨ��Ũ���ᣬ��ȥH2O��������d�������ռ��������������������װ�����Ӻú���Ҫ���еIJ����Ǽ��װ�õ������ԣ���4����֤�����к���FeO�����������������ӵĻ�ԭ�ԣ��������Լ�������ʵ�����Ϊ��ȡ�����������ӹ���ϡ�����ܽ⣬Ȼ������Һ�м�����KMnO4��Һ������Һ��ɫ��ȥ����˵�������к���FeO��
���㣺���黯ѧʵ������������������Ʊ����������ռ���ʵ�鷽������������ۣ����ӷ���ʽ����д��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д� ���ۻ������ؿ���Ϊҩ���������Ƽ��Ե�����Ⱦ�����Ը��ȼ�������ṹ��ʽ���£����жԼ��ۻ������ص�����������ȷ���ǣ�������
���ۻ������ؿ���Ϊҩ���������Ƽ��Ե�����Ⱦ�����Ը��ȼ�������ṹ��ʽ���£����жԼ��ۻ������ص�����������ȷ���ǣ�������| A�����ۻ������ؿ��Է�����ȥ��Ӧ |
| B��1 mol���ۻ������ؿ��� 6 molH2�����ӳɷ�Ӧ |
| C�����ۻ������ؿ��� NaOH ��Һ�ڼ��������·���ˮ�ⷴӦ |
| D�����ۻ������ؼ�����Na��Ӧ����H2��������NaHCO3��Ӧ����CO2 |
 Na2S2O3(aq)
Na2S2O3(aq)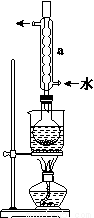
 ��2I��
��2I��