��Ŀ����
����Ҫ���û�ѧ�����ʾ�����йط�Ӧ��
��1����������������ʴʱ�������ĵ缫��Ӧʽ�� ��
��2����ʯī�Ӹ�����ͭ����������������ͭ��Һʱ�������ĵ缫��Ӧʽ�� ��
��3����ʯī���缫��ⱥ��ʳ��ˮ�Ļ�ѧ����ʽ�� ��
��4������Ũ�ȡ��������NaHSO4��Һ��Ba��OH��2��Һ��ϣ���Ӧ�����ӷ���ʽ�� ��
��5��0.3molB2H6������������ȼ�����ɹ�̬�����������Һ̬ˮ���ų�649.5kJ������д���÷�Ӧ���Ȼ�ѧ����ʽ�� ��
��1����������������ʴʱ�������ĵ缫��Ӧʽ��
��2����ʯī�Ӹ�����ͭ����������������ͭ��Һʱ�������ĵ缫��Ӧʽ��
��3����ʯī���缫��ⱥ��ʳ��ˮ�Ļ�ѧ����ʽ��
��4������Ũ�ȡ��������NaHSO4��Һ��Ba��OH��2��Һ��ϣ���Ӧ�����ӷ���ʽ��
��5��0.3molB2H6������������ȼ�����ɹ�̬�����������Һ̬ˮ���ų�649.5kJ������д���÷�Ӧ���Ȼ�ѧ����ʽ��
���㣺�缫��Ӧ�͵�ط�Ӧ����ʽ,���ӷ���ʽ����д,�Ȼ�ѧ����ʽ
ר�⣺
��������1����������������ʴʱ�������������õ��ӷ�����ԭ��Ӧ��
��2��Cu�������������ͭ��Һʱ��������Cuʧ���ӷ���������Ӧ��
��3����ʯī���缫����Ȼ�����Һʱ�������������ӷŵ����������������������ӷŵ�����������ͬʱ��Һ�л�����NaOH��
��4�������ʵ�����NaHSO4��Ba��OH��2��Ӧ�������ᱵ��NaOH��ˮ��
��5��0.3molB2H6������������ȼ�����ɹ�̬�����������Һ̬ˮ���ų�649.5kJ��������1molB2H6��ȫ��Ӧ�ų�2165 kJ�������ݴ���д�Ȼ�ѧ��Ӧ����ʽ��
��2��Cu�������������ͭ��Һʱ��������Cuʧ���ӷ���������Ӧ��
��3����ʯī���缫����Ȼ�����Һʱ�������������ӷŵ����������������������ӷŵ�����������ͬʱ��Һ�л�����NaOH��
��4�������ʵ�����NaHSO4��Ba��OH��2��Ӧ�������ᱵ��NaOH��ˮ��
��5��0.3molB2H6������������ȼ�����ɹ�̬�����������Һ̬ˮ���ų�649.5kJ��������1molB2H6��ȫ��Ӧ�ų�2165 kJ�������ݴ���д�Ȼ�ѧ��Ӧ����ʽ��
���
�⣺��1����������������ʴʱ�������������õ��ӷ�����ԭ��Ӧ�������������ӣ��缫��ӦʽΪ2H2O+O2+4e-=4OH-���ʴ�Ϊ��2H2O+O2+4e-=4OH-��
��2��Cu�������������ͭ��Һʱ��������Cuʧ���ӷ���������Ӧ����ͭ���Ӷ��ܽ⣬�缫��ӦʽΪCu-2e-=Cu2+���ʴ�Ϊ��Cu-2e-=Cu2+��
��3����ʯī���缫����Ȼ�����Һʱ�������������ӷŵ����������������������ӷŵ�����������ͬʱ��Һ�л�����NaOH����ط�ӦʽΪ2NaCl+2H2O
2NaOH+H2��+Cl2�����ʴ�Ϊ��2NaCl+2H2O
2NaOH+H2��+Cl2����
��4�������ʵ�����NaHSO4��Ba��OH��2��Ӧ�������ᱵ��NaOH��ˮ�����ӷ���ʽΪBa2++OH-+H++SO42-=BaSO4��+H2O���ʴ�Ϊ��Ba2++OH-+H++SO42-=BaSO4��+H2O��
��5��0.3molB2H6������������ȼ�����ɹ�̬�����������Һ̬ˮ���ų�649.5kJ��������1molB2H6��ȫ��Ӧ�ų�2165 kJ����������Ȼ�ѧ��Ӧ����ʽΪB2H6��g��+3O2��g��=B2O3��s��+3H2O��l����H=-2165 kJ?mol-1 ���ʴ�Ϊ��B2H6��g��+3O2��g��=B2O3��s��+3H2O��l����H=-2165 kJ?mol-1 ��
��2��Cu�������������ͭ��Һʱ��������Cuʧ���ӷ���������Ӧ����ͭ���Ӷ��ܽ⣬�缫��ӦʽΪCu-2e-=Cu2+���ʴ�Ϊ��Cu-2e-=Cu2+��
��3����ʯī���缫����Ȼ�����Һʱ�������������ӷŵ����������������������ӷŵ�����������ͬʱ��Һ�л�����NaOH����ط�ӦʽΪ2NaCl+2H2O
| ||
| ||
��4�������ʵ�����NaHSO4��Ba��OH��2��Ӧ�������ᱵ��NaOH��ˮ�����ӷ���ʽΪBa2++OH-+H++SO42-=BaSO4��+H2O���ʴ�Ϊ��Ba2++OH-+H++SO42-=BaSO4��+H2O��
��5��0.3molB2H6������������ȼ�����ɹ�̬�����������Һ̬ˮ���ų�649.5kJ��������1molB2H6��ȫ��Ӧ�ų�2165 kJ����������Ȼ�ѧ��Ӧ����ʽΪB2H6��g��+3O2��g��=B2O3��s��+3H2O��l����H=-2165 kJ?mol-1 ���ʴ�Ϊ��B2H6��g��+3O2��g��=B2O3��s��+3H2O��l����H=-2165 kJ?mol-1 ��
���������⿼�����ӷ���ʽ���Ȼ�ѧ����ʽ���缫��Ӧʽ����д�����ؿ���ѧ���Ի������۵����⣬ע����д���ص缫��Ӧʽʱ���ж��������ϣ�����д�缫��Ӧʽ��ע�⣨4���з�Ӧ�������д���ӷ���ʽ��Ϊ�״��㣮

��ϰ��ϵ�д�
�����Ŀ
���й��ڷ�Ӧ������˵����ȷ���ǣ�������
| A��Zn��s��+CuSO4��aq���TZnSO4��aq��+Cu��s����H=-216 kJ/mol����Ӧ���������������������� |
| B����ͬ�����£����1 mol��ԭ�������е�����ΪE1��1 mol ����ӵ�����ΪE2����2E1��E2 |
| C��101 kPaʱ��2H2��g��+O2��g���T2H2O��g����H=-483.6 kJ/mol��H2��ȼ���ȡ�H=-241.8 kJ/mol |
| D��H+��aq��+OH-��aq���TH2O��l����H=-57.3 kJ/mol����1 mol NaOH������������Һ�뺬0.5 mol H2SO4��Ũ�����Ϻ�ų�57.3 kJ ������ |
��������������ǣ�������
| A��10ml��������Ϊ98%��H2SO4����10mlˮϡ�ͺ�H2SO4��������������49% |
| B������0.1mol/L��Na2CO3��Һ480ml������500ml����ƿ |
| C���ڱ���£���22.4L��������1Lˮ�У��õ�1mol/L�İ�ˮ |
| D����2�ȷݲ����͵��ռ���Һ�зֱ����һ������Na2O2��Na2O��ʹ��Һ��ǡ�ñ��ͣ�������Na2O2��Na2O�����ʵ���֮�ȵ���1��1�������¶Ȳ��䣩 |
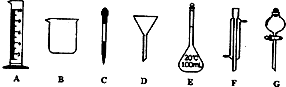
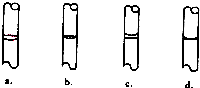
 A��B��C��D��E����ѧ������5�ֻ����A��B�������Ԫ��X��Y�ĵ����������г����Ľ�����������ʼ�Ĺ�ϵ��ͼ��ʾ��
A��B��C��D��E����ѧ������5�ֻ����A��B�������Ԫ��X��Y�ĵ����������г����Ľ�����������ʼ�Ĺ�ϵ��ͼ��ʾ��