��Ŀ����
������ѧ�����ʣ���ѧ��ʵ���������ʵ���� ��ϴ�ӡ���ȷ�����¹��ڶԳ����������ϴ�ӵ�˵���������
A. ϴ�ӵ�Ŀ��һ���dz�ȥ���������������Ե����ʣ�����߹���Ĵ���
B. ϴ�ӵIJ����ǣ����������Ĺ���ֱ��ע��ϴ�Ӽ���û���壬��ϴ�Ӽ���Ȼ���¼���
C. ϴ�ӵ��Լ�������Ҫһ���ѡ������ˮ����ˮ���Ҵ�������ʵı�����Һ
D. �Ƿ�ϴ���ļ��飺ȡ���һ��ϴ��Һ���������γ���Ӧ�����ĸ���Һ�е�����
��ϰ��ϵ�д�
�����Ŀ




 NH2OH ��
NH2OH ��
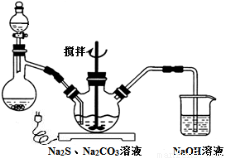
 pC(s)+qQ(g)��m��n��p��qΪ������ʱ���ﵽƽ��ı�־�ǣ�
pC(s)+qQ(g)��m��n��p��qΪ������ʱ���ﵽƽ��ı�־�ǣ�