��Ŀ����
Ϊ�ⶨ������ͭ�����к���������������������������ʣ���Ʒ��CuO����������������ȡ�������弰�ⶨ���������о���ˮ�ĺ�����ij��ѧ�С�����������ʵ�飺
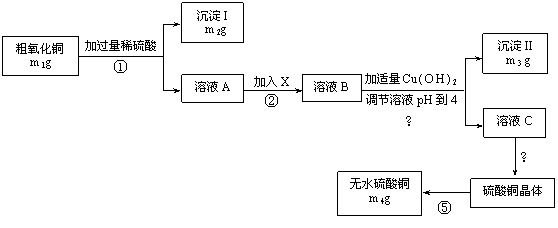 |
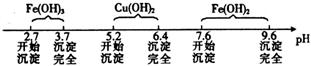
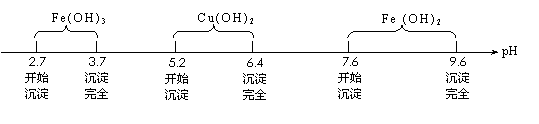 ��֪Fe3����Cu2����Fe2�� ����������ˮ��Һ���γ��������������pH��Χ����ͼ��ʾ��
��֪Fe3����Cu2����Fe2�� ����������ˮ��Һ���γ��������������pH��Χ����ͼ��ʾ��
��ش��������⣺
��1��������ҺpH�ܷ�ѡ��NaOH��Һ��˵������
��2������IIΪ ���������ʵ�����Ϊ
��3����������XĿ����
��4���ڲⶨ���õ�����CuSO4��xH2O���нᾧˮxֵ��ʵ������У����ⶨ���xֵƫ���ܵ�ԭ���� �� ��
a �����¶ȹ��� b ���ȵ�������ʱ�о���������н���
c ���Ⱥ���ڿ�������ȴ
��5����Ʒ��CuO����������Ϊ ���ú�m�Ĵ���ʽ��ʾ����
(1)���ܣ���Ϊ��������Na+��(2��) (2)Fe��OH��3 (2��) �����ᾧ(2��)
(3)��Fe2+������Fe3+(2��) (4)a b (2��) (5) 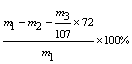 (2��)
(2��)




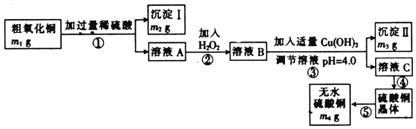
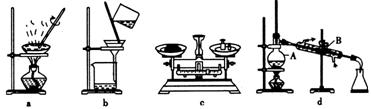
 ��ȡ��ˮ����ͭ��ij��ѧ�С�����������ʵ�飺
��ȡ��ˮ����ͭ��ij��ѧ�С�����������ʵ�飺




