��Ŀ����
��ð��һ�ֳ����IJ����˻��˸�ð����ԡ����顢���գ���ȿ�ˮ����Ϣ���ʵ�����һЩ���Ƹ�ð��ҩ�������滺֢״�����Ს�飮
��1��ҩ�ﰢ˾ƥ����һ�ִ�ͳ�����Ƹ�ð���յ�ҩ���ӷ��ӽṹ�Ͽ��������Կ���������������ṹ��ʽΪ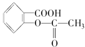 ����Ϊһ�ֽ�����ʹҩ�����ڸ�ð���գ����нϺõ���Ч������������ڿո����ã�����θ���������̼���ԭ���ǰ�˾ƥ����θ�лᷢ������ˮ�ⷴӦ�����ɣ��ýṹ��ʽ��ʾ�� �� ����˾ƥ�ַ���ˮ����ܵ���������ӣ�H+���Ĺ�������ԭ���� ������������˶�θ�����Ĵ̼���
����Ϊһ�ֽ�����ʹҩ�����ڸ�ð���գ����нϺõ���Ч������������ڿո����ã�����θ���������̼���ԭ���ǰ�˾ƥ����θ�лᷢ������ˮ�ⷴӦ�����ɣ��ýṹ��ʽ��ʾ�� �� ����˾ƥ�ַ���ˮ����ܵ���������ӣ�H+���Ĺ�������ԭ���� ������������˶�θ�����Ĵ̼���
��2��Ϊ���ٸ����ã�һ���˷����ҩ����ͬʱ����������ҩ��ڢ�����þ����������������̼����У�ѡȡ��һ�֣� ��
��1��ҩ�ﰢ˾ƥ����һ�ִ�ͳ�����Ƹ�ð���յ�ҩ���ӷ��ӽṹ�Ͽ��������Կ���������������ṹ��ʽΪ
 ����Ϊһ�ֽ�����ʹҩ�����ڸ�ð���գ����нϺõ���Ч������������ڿո����ã�����θ���������̼���ԭ���ǰ�˾ƥ����θ�лᷢ������ˮ�ⷴӦ�����ɣ��ýṹ��ʽ��ʾ��
����Ϊһ�ֽ�����ʹҩ�����ڸ�ð���գ����нϺõ���Ч������������ڿո����ã�����θ���������̼���ԭ���ǰ�˾ƥ����θ�лᷢ������ˮ�ⷴӦ�����ɣ��ýṹ��ʽ��ʾ����2��Ϊ���ٸ����ã�һ���˷����ҩ����ͬʱ����������ҩ��ڢ�����þ����������������̼����У�ѡȡ��һ�֣�
���㣺�л���Ľṹ������
ר�⣺�л���Ļ�ѧ���ʼ��ƶ�
��������1����˾ƥ�ֵĽṹ��ʽΪ ����-COOC-���ɷ���ˮ�⣬����������ˮ�����������ԣ�
����-COOC-���ɷ���ˮ�⣬����������ˮ�����������ԣ�
��2��Ϊ���ٸ����ã����������ӷ�Ӧ���Ҳ��������ж����ʣ�
 ����-COOC-���ɷ���ˮ�⣬����������ˮ�����������ԣ�
����-COOC-���ɷ���ˮ�⣬����������ˮ�����������ԣ���2��Ϊ���ٸ����ã����������ӷ�Ӧ���Ҳ��������ж����ʣ�
���
�⣺��1����˾ƥ�ֵĽṹ��ʽΪ ����-COOC-���ɷ���ˮ�⣬ˮ�ⷴӦΪ
����-COOC-���ɷ���ˮ�⣬ˮ�ⷴӦΪ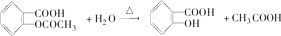 ��ˮ�����
��ˮ�����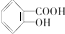 ��CH3COOH�����ᣬ�ܵ���������ӵĹ�������ԭ����3���������˶�θ�����Ĵ̼���
��CH3COOH�����ᣬ�ܵ���������ӵĹ�������ԭ����3���������˶�θ�����Ĵ̼���
�ʴ�Ϊ�� ��CH3COOH��
��CH3COOH��
��2��Ϊ�˼����丱���ã�����������þ��û�н�����ȣ���������������Ȼ��������ȣ��������ж��������ӣ�������̼��Ƽȼ��ٸ������ֲ���ƣ����ѡ��̼��ƣ�����CaCO3+2H+�TCa2++CO2��+H2O��Ч����ã��ʴ�Ϊ��̼��ƣ�
 ����-COOC-���ɷ���ˮ�⣬ˮ�ⷴӦΪ
����-COOC-���ɷ���ˮ�⣬ˮ�ⷴӦΪ ��ˮ�����
��ˮ����� ��CH3COOH�����ᣬ�ܵ���������ӵĹ�������ԭ����3���������˶�θ�����Ĵ̼���
��CH3COOH�����ᣬ�ܵ���������ӵĹ�������ԭ����3���������˶�θ�����Ĵ̼����ʴ�Ϊ��
 ��CH3COOH��
��CH3COOH����2��Ϊ�˼����丱���ã�����������þ��û�н�����ȣ���������������Ȼ��������ȣ��������ж��������ӣ�������̼��Ƽȼ��ٸ������ֲ���ƣ����ѡ��̼��ƣ�����CaCO3+2H+�TCa2++CO2��+H2O��Ч����ã��ʴ�Ϊ��̼��ƣ�
���������⿼���л���Ľṹ�����ʣ�Ϊ��Ƶ���㣬���չ����������ʵĹ�ϵΪ���Ĺؼ������ط�����ҩ�����õ��ۺ�����Ŀ��飬ע��������ѧ�����л���ѧ֪ʶȥ�������Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 ���ƽ̸�������ѡ����ĩ���100��ϵ�д�
���ƽ̸�������ѡ����ĩ���100��ϵ�д�
�����Ŀ
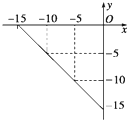
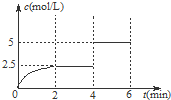 һ���¶����п��淴Ӧ��A��g��+2B��g��?2C��g��+D��g�����ֽ�4mol A��8mol B�������Ϊ2L��ij�ܱ������У���Ӧ��4minʱ���ı�ijһ���������C�����ʵ���Ũ����ʱ��仯��������ͼ��ʾ�������й�˵������ȷ���ǣ�������
һ���¶����п��淴Ӧ��A��g��+2B��g��?2C��g��+D��g�����ֽ�4mol A��8mol B�������Ϊ2L��ij�ܱ������У���Ӧ��4minʱ���ı�ijһ���������C�����ʵ���Ũ����ʱ��仯��������ͼ��ʾ�������й�˵������ȷ���ǣ�������| A��0��2 min�ڣ�����Ӧ���������� |
| B��4 minʱ��A��ת����Ϊ50% |
| C��6 minʱ��B���������Ϊ25% |
| D��4 minʱ���ı�������ƽ�����淴Ӧ�����ƶ� |
�����Ƕ�Ԫ���ᣬ���������Һ�����ԣ���0.01mol?L-1KHC2O4��Һ�У����й�ϵ��ȷ���ǣ�������
| A��c ��K+��+c ��H+��=c ��HC2O4-��+c ��OH-��+c ��C2O42-�� |
| B��c ��C2O42-����c ��H2C2O4�� |
| C��c ��HC2O4-��+c ��C2O42-��=0.1 mol?L-1 |
| D��c ��K+��=c ��H2C2O4��+c ��HC2O4-��+c ��C2O42-�� |
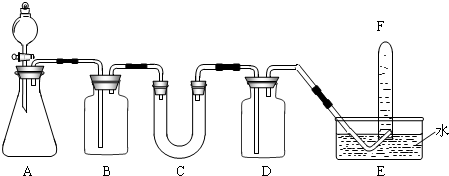
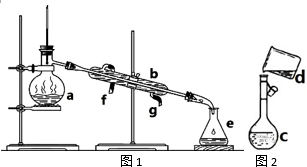 �����������ơ���װ��ʹ�÷�������ѧ��ѧʵ��Ļ�������ͼΪ����ʵ��װ�ã�
�����������ơ���װ��ʹ�÷�������ѧ��ѧʵ��Ļ�������ͼΪ����ʵ��װ�ã�