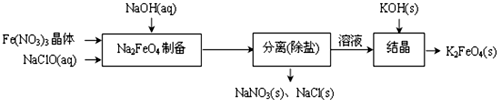
��Ŀ����
����ָ����Ӧ�����ӷ���ʽ��ȷ���ǣ�������
| A��������Һ��ͨ��������CO2��C6H5O-+CO2+H2O��C6H5OH+HCO3- | ||||
| B��FeS�������ϡ������Һ�У�FeS+2H+=Fe2++H2S�� | ||||
| C����NH4��2Fe��SO4��2��Һ�����NaOH��Һ��Ӧ��Fe��OH��2��Fe2++2OH-=Fe��OH��2�� | ||||
D���ö��Ե缫����Ȼ�����Һ��2Cl-+2H+
|
���㣺���ӷ���ʽ����д
ר�⣺
������A���������������̼��Ӧ���ɱ�����̼�����ƣ�
B���������ǿ�������ԣ��ܹ����������������������ӣ�
C��©���˰������������������ӵķ�Ӧ��
D��ˮΪ������ʣ�Ӧ������ѧʽ��
B���������ǿ�������ԣ��ܹ����������������������ӣ�
C��©���˰������������������ӵķ�Ӧ��
D��ˮΪ������ʣ�Ӧ������ѧʽ��
���
�⣺A��������Һ��ͨ��������CO2�����ӷ���ʽΪ��C6H5O-+CO2+H2O��C6H5OH+HCO3-����A��ȷ��
B��FeS�������ϡ�����У��������Ӻ������Ӷ��ܹ���������������������������Ӧ�����ӷ���ʽΪFeS+4H++3NO3-=Fe3++SO42-+3NO��+2H2O����B����
C����NH4��2Fe��SO4��2��Һ�����NaOH��Һ�����ӷ���ʽΪ��2NH4++2OH-+Fe2++2OH-=Fe��OH��2��+2NH3?H2O����C����
D���ö��Ե缫����Ȼ�����Һ�����ӷ���ʽ��2Cl-+2H2O
H2��+Cl2��+2OH-����D����
��ѡ��A��
B��FeS�������ϡ�����У��������Ӻ������Ӷ��ܹ���������������������������Ӧ�����ӷ���ʽΪFeS+4H++3NO3-=Fe3++SO42-+3NO��+2H2O����B����
C����NH4��2Fe��SO4��2��Һ�����NaOH��Һ�����ӷ���ʽΪ��2NH4++2OH-+Fe2++2OH-=Fe��OH��2��+2NH3?H2O����C����
D���ö��Ե缫����Ȼ�����Һ�����ӷ���ʽ��2Cl-+2H2O
| ||
��ѡ��A��
���������⿼�������ӷ���ʽ�������жϣ����շ����ķ�Ӧ�����ӷ�Ӧ����д����Ϊ���Ĺؼ�����Ŀ�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
NaHCO3��Һ�������еķ��Ӻ������У�������
��NaHCO3 ��H2O ��H2CO3 ��HCO3- ��H+ ��OH-��CO32-��
��NaHCO3 ��H2O ��H2CO3 ��HCO3- ��H+ ��OH-��CO32-��
| A���٢ڢ� |
| B���ڢܢݢ� |
| C���ڢۢܢݢޢ� |
| D���٢ڢۢܢݢޢ� |
��Ӧ2NO+2CO
N2+2CO2��Ӧ��������β���ľ����������ж���ȷ���ǣ�������
| ||
| A���÷�Ӧ���û���Ӧ |
| B���÷�Ӧ�Ǹ��ֽⷴӦ |
| C��CO�������� |
| D��NO������ԭ��Ӧ |
�����Լ����࣬���л������ǻ۵Ľᾧ�����г������ı仯���ڻ�ѧ�仯���ǣ�������
| A����ʯ�ɽ� | B��ˮ��ʯ�� |
| C����ˮ�ɱ� | D�����Ƽ��� |
�������ʼ������������л�ԭ�Ե��ǣ�������
| A��Fe |
| B��FeCl2 |
| C��Fe3+ |